This document is reprinted from the original at http://www.savina.com/confluence/hormone/m2f/index.htm
This document can no longer be found at its original site, so this is just a copy to keep it alive, and please note, it is at least ten years old now, so the information from it should be taken with a pinch of salt!
Abstract
This document contains a list of frequently asked questions and their answers regarding hormone therapy (secondary sexual reassignment) for male-to-female transsexuals. More generally, this document contains information about gonadal hormones and anti-hormones, so it can be a helpful reference for the treatment of androgen and estrogen-sensitive conditions--for example, certain cancers of the reproductive organs and breasts.
Sponser Advertizement Space
The hormone faq is viewed or scanned about 6,000 times per week. To help offset research and publishing costs, for U.S. $20/month, or $100/6 months, you can rent billboard/link space in the F2M or M2F version of this document to advertize certain directories, services or products to transsexuals. Contact confluence at savina dot com (Confluence Publications).
Location and Updates
The authoritative location of this document is - http://www.savina.com/confluence/hormone/m2f/
Large (~209Kb) flat file suitable for download and offline browsing - http://www.savina.com/confluence/hormone/m2f/flat.htm
Ftp service has been discontinued due to the relatively low demand (less than 0.5% of web traffic) and the inconvenience of maintaining asciified tables.
Each page in this document has a footer displaying the date of the most recent change.
Please send additions, corrections, and suggestions to confluence at savina dot com.
Copyright and Terms of Redistribution
Contents copyright © 1994-1998 by confluence at savina dot com (Confluence Publications). Permission is granted to freely redistribute, provided that:
- The entire document from first line of header to last line of footer is reproduced unabridged, unedited (except for layout format) and you notify Confluence Publications, or;
- Less than 20 lines are quoted to answer a specific question, and the authoritative location of this document is specified in addition to the quote.
- Redistribution of the web version must be via a faithful mirror of all document components, updated monthly. Other public cybernetic redistribution must also be updated monthly. This will help ensure that the latest, safest information is available to those who seek it. This document is hosted on a stable server with fast, redundant network connections. You are strongly encouraged to link directly to http://www.savina.com/confluence/hormone/m2f/ instead of redistributing a copy.
- The republished document or quote is made available without charge (beyond a nominal charge for accessing e-mail, public net news, public web sites, and non-profit support groups). Hardcopy or cybernetic redistribution of any portion for profit requires negotiation with Confluence Publications.
All other rights are reserved.
Disclaimers
The answers in this document are collected from a variety of sources: medical literature, pharmaceutical company advertizement, verbal advice of medical doctors, second-hand anecdotes, and personal experience. Despite the authoritative tone of this document, it is presented for educational interest only, not direct advice. It contains opinions, sweeping generalizations, and at least one mistake. The author is not a medical doctor, and makes no claim or warranty as to the suitability of the information in this document for application to any particular individual. YOU, the reader, take sole responsibility for interpretation and application of this information. Form your own opinions by doing your own research. May your favorite deity curse you if you seriously consider sueing the author for misinforming you.
The endocrine feedback system is intricate, delicate, and poorly understood. Even the experts do not entirely agree on how to best meddle with it. Hormone therapy is fraught with risk as well as promise. Be sure you have fully considered the implications before you start. Work with a medical doctor who is qualified to interpret your signs, symptoms, blood tests, and development in the context of your personal medical history. Do not take hormones that you did not obtain directly from a licensed pharmaceutical distributor; the quality of drugs obtained through other channels is not only suspect, but possibly dangerous--especially those in injectable form.
Notes
- The words "female" and "male" refer to the original physical form, not to gender identification.
- This document does not address hormone therapy of the individual with an endocrine system disorder.
- Pre-op hormone dosages are determined mainly from verbal advice of medical doctors, second-hand anecdotes, and personal experience.
- Post-op hormone dosages are determined mainly from the Physician's Desk Reference (PDR) according to the recommendation of hormone replacement of normal gonadal hormone production.
- Drugs are ranked by the following criteria:
– Safety and efficacy: excellent, good, fair, unknown, or poor. This is based on normal indications (i.e., when the primary indication is female hormone replacement, it is suitable for transsexual hormone therapy), delivery method (sustained-release injectibles and transdermal films are easier on the liver than oral tablets), literature, medical doctor advice, anecdotes, and other factors noted in the individual hormone comment field. Safety and efficacy are generally closely linked, so the ranking is combined for all drugs except anti-androgens. – Source: unknown, animal by-products, or live animals
- Of the injectable hormones and anti-hormones available, only those that are sustained-release (requiring injection less frequently than once per week) are listed.
- The adverse effects listed in this document are gleaned primarily from drug information sheets and the PDR. They are translated from medibabble into English where possible. While this information should not be taken lightly, it should be viewed with slight suspicion, since it is first and foremost advertizement and legal copy from pharmaceutical companies. To attempt to reduce their exposure to lawsuits, they list not only the effects reasonably shown to be caused by the drug during clinical trial(s), but also every other adverse effect that the patients experienced while taking the drug--or any other drug of the same class--whether or not the effect was proven statistically relevant by controlled study for the drug in question. In particular, the reader should not be unduly worried about the mention of increase of body hair and loss of scalp hair from estrogens, nor about increase of body hair and deepening of voice from androgen receptor antagonists and GnRH agonists. Finally, adverse effects are only listed here if they make sense in their application to transsexuals, i.e., adverse effects on uniquely female organs are not listed for drugs intended for male-to-female transsexuals, and vice-versa. One should really read the PDR for the drugs of interest in order to provide context for the adverse effects listed in this document.
Questions Answered in This Document
- What are hormones, and how do they work?
- What are normal endogenous androgen and estrogen levels levels?
- What effecteffect does contrasexual hormone therapy have, and how soon?
- What are the popular treatment philosophies?
- How are hormones delivered?
- How can the intended effects of hormone therapy be maximised and the dangers minimized?
- How can one obtainobtain hormones?
- Are birth-control pills a good source of estrogen?
- Exactly what hormones are available? What are the details on popularity, dosage, availability, contraindications, adverse effects, etc.?
- Where is there more information about hormones?
What Are Hormones, and How Do They Work?
Hormones are long-range chemical messengers of the body, manufactured and controlled by the endocrine system. Hence the title of endocrinologist for hormone doctors.
The hypothalamus produces gonadotropin-releasing hormone (GnRH). This signals the anterior pituitary gland to synthesize and release luteinizing hormone (LH). To a lesser degree, GnRH also triggers the synthesis and release of follicle stimulating hormone (FSH). Subsequently, LH and FSH signal the gonads (ovaries in females, testes in males) to synthesize and release hormones that cause differentiation of the body tissue into female or male form: estrogens, progesterones, and testosterones. A small quantity of testosterones are also produced by the adrenal gland. Proportionally, females have more estrogens and progesterones than males; males have more testosterones.
Estrogens include natural and synthetic estradiols, estrones and estriols. They excite estrogenic receptors, causing the body to differentiate into female form and function. Natural and synthetic estrogens are hereafter referred to simply as estrogens.
Progestogens (or progestins) are synthetic progesterone analogues. Progesterones and progestogens excite progesteronic receptors, which in cooperation with estrogenic activity, cause the body to further differentiate into female form and function. Natural and synthetic progesterones are hereafter referred to simply as progesterones.
Various testosterones are collectively known as androgens. They excite androgenic receptors, causing the body to differentiate into male form and function. Natural and synthetic testosterones are hereafter referred to simply as androgens.
Anti-hormones can be useful in transsexual hormone therapy because they block hormone action or production. The basic mechanisms are:
- Androgen receptor antagonist: blocks the action of androgens at certain receptor sites.
- Androgen conversion inhibitor: blocks the conversion of one type of androgen to another.
- GnRH agonist: briefly overstimulates then effectively suppresses pituitary LH and FSH production.
Aggressive exogenous hormone therapy indirectly reduces endogenous (natural) gonadal hormone production by fooling the pituitary into thinking that there are plenty of hormones already in the body; consequently, the pituitary reduces the LH and FSH signals that stimulate the gonads.
Postnatally administered hormones do not cause development of are opposite those born with. However, postnatal contrasexual hormone therapy does cause development of secondary sex characteristics as described below.
What Are Normal Endogenous Androgen and Estrogen Levels?
The normal endogenous androgen range in a male is 300-1100 nanograms per deciliter. Estrogen is generally below 50 picograms per milliliter.
The normal endogenous androgen range in a female is 10-100 nanograms per deciliter. (within this range lower numbers are not necessarily considered better.) There are dramatic cyclic and individual variations of estrogen (estradiol + estrone), with 100-400 picograms per milliliter being the most usual, with 25-700 being possible depending on the individual. 400 is considered a nominal "mid-peak" (ovulation) level. 200-250 is considered a reasonable target for exogenous estrogen treatment. Note that only natural estrogens can be meaningfully measured, so it you take any estrogen besides estradiol valerate, estradiol cypionate, or estradiol, you will not be able to accurately judge the results of a blood test.
What effect does female hormone therapy have on a male, and how soon?
The longer after puberty hormone therapy is started, the less effective it is--but not a linear scale, e.g., results are considerably more dramatic in an 18 year old than a 28 year old, but results are not on the average dramatically different between a 38 year old and a 48 year old.
The following effects have been observed in varying degrees--anywhere from little to moderate--with extended treatment. With effective and continuous dosages, most of the changes that a particular body is genetically prone to start within 2 to 4 months, start becoming irreversible within 6 to 12 months, start leveling off somewhat within 2 years, and be mostly done within 5 years. The leveling may take longer if the testes are not removed. High levels of estrogen will cause faster development up to a point, but not better results in the long term than moderate levels of estrogen.
-
Fertility decreases. Sperm count drops rapidly. Sometimes it returns to almost normal if hormonal treatment is discontinued within the first couple of months, but permanent sterility can occur in as little as six months. This should not be counted on for birth control, because a miniscule sperm count might remain until the testes are surgically removed. Estrogens, progesterones, and gonadal androgen production inhibitors are the chemicals responsible for lowering fertility. It appears to the author that the other types of anti-androgens do not necessarily effect fertility--but one would be wise to take frequent fertility tests if one chooses to employ only the other types of anti-androgens with the intent of maintaining fertility.
-
Male sex drive decreases. Directly stimulated erections can become infrequent and difficult to maintain. Spontaneous erections usually stop. Semen secretion decreases, usually resulting in less intense ejeculatory orgasms (however, the ability to achieve a satisfying orgasm--even with little or no semen--is determined more by psychological factors and frequent practice than anything else). The testes and prostate atrophy. The penile skin also shrinks if erections are not regularly encouraged.
-
Breast size increases. Typical growth is one to two cup sizes below closely related females (mother, sisters). The growth is not always symmetrical--neither is it for females. Sometimes the areoles and nipples swell, but generally not significantly, unless the body is less than a decade past puberty.
-
Fat is redistributed. The face becomes more typically female in shape. Fat tends to move away from the waist and toward the hips and buttocks.
-
Body hair growth (not including head, face, or pubic area) slows, becomes less dense, and may lighten in color.
Many people also report the following effects, but they are not verified in any medical literature that the author has read:
- If exercise is not increased, some muscle tone is lost.
- Outer skin layer becomes thinner, lending a finer translucent appearance and increased susceptibility to scratching and bruising. Tactile sensation becomes more intense.
- Oil and sweat glands become less active, resulting in dryer skin, scalp, and hair.
- Scalp hair becomes thicker, and male pattern baldness generally stops advancing. In some cases, a fine fuzz may grow back along the line of where scalp hair was recently lost--but only from the living follicles, not dead ones.
- Metabolism decreases. Given a caloric intake and exercise regimen consistent with pre-hormonal treatment, one tends to gain weight, lose energy, need more sleep, and become cold more easily. Sometimes the ability to concentrate is also initially diminished, but the tiredness and distraction generally pass once the body and brain become used to operating with less androgens to maintain intensity.
- Fingernails become thinner and more brittle.
- Body odors (skin and urine) change. They become less "tangy" or "metallic" and more "sweet" or "musky".
- Internal emotions are amplified, becoming more apparent, distinguishable, and influential. Some people report reduced anxiety and increased sense of well-being. This could be a placebo effect. Changing the hormone therapy (adjusting dosages up or down in the regimen) sometimes causes a week or two of depression and otherwise unexplainable emotional angst.
- "Female" sex drive and enjoyment increase. This observation is obviously completely subjective since males have no way to directly compare the experience. Non-ejeculatory orgasms become more likely for those with the predisposition to have them, if for no other reason than the fact that ejeculatory orgasms are difficult or impossible to achieve, and the need for sexual release forces a rewiring of perceptions and responses.
Female hormones do not:
- Cause the voice to increase in pitch.
- Dramatically reduce facial hair growth in most people. There are some exceptions with people who have the proper genetic predisposition and/or are less than a decade past puberty.
- Change the shape or size of bone structure. However, they may change the bone density slightly.
What Are the Popular Treatment Philosophies?
The following estrogen dosage philosophies are popular for treatment of male-to-female transsexuals, and are presented in descending order of preference in the humble opinion of the author.

Clearly, philosophy 1 and 2 reasonings flatly contradict each other. There are good endocrinologists in each camp, which demonstrates that we still really do not know exactly how hormones work. However, there is more compelling evidence for the reasoning of philosophy 1. In some people philosophy 1 might have a not have quite as steep of a ramp of results as philosophy 2--but, with patience, the results seem to be just as good. The A philosophies adjust to the body's assimilation of the estrogens, whereas the B philosophies assume "one size fits all." Gross empirical results, e.g., breast development, should be used as a secondary rather than primary indicator. Finally, note that the endogenous level of estrogen in females (F2M) seems to be a less important factor for development than the endogenous level of androgens in males (M2F).
The following estrogen coadministration philosophies are popular for treatment of male-to-female transsexuals, and are presented in descending order of preference in the humble opinion of the author.

How Are Hormones Delivered?

Note that the absorbtion of oral preparations varies greatly among individuals. With some the absorbtion is poor; in that case, injection or transdermal film is indicated.
Sustained-release intramuscular injectable hormones are suspended in oil. This is the usual procedure for administration:
- If you are very sensitive to pain, obtain 2 new needles for each administration: 1 to fill the syringe (18-22 guage), and another for the injection (22 guage). That way the injection needle will be entirely sharp. Be careful not to drag the injection needle across anything, even skin, before the injection, because that will dull it.
- If you are fairly tolerant of pain, or cannot afford 2 needles for each injection, then use the same new needle (22 guage) to fill the syringe as to make the injection. Do not under any circumstances reuse needles between injection periods, or between different people.
- Warm the vial (ampule) between your hands for a moment to help the oil flow more freely.
- Cleanse the top of the vial and the area for injection with a swipe of povidone-iodine (10%), or if you cannot obtain that, use rubbing alcohol (95-99%) or hydrogen peroxide (3-5%).
- The best intramuscular injection sites are the upper outer quadrant of the buttock, or upper outer thigh. Either is fine, as long as you are hitting at least two inches of fat and muscle, not bone or an artery.
- Securely mount the drawing needle on the syringe, then pull back the plunger about 1/4-1/3 cc farther than the intended injection amount (e.g., if you intend to inject 1 cc, then draw back 1 1/4 - 1 1/3 cc of air).
- With the vial right-side-up, insert the needle in the top, such that the needle end is in the bottle air, but not the oil. Inject all of the air from the syringe into the vial.
- Turn everything so the vial is upside-down, be sure the needle end is in oil (not air, and not bumping against the glass), then slowly but firmly draw back the plunger until you have a bit more than the injection amount. You will probably see some small air bubbles; that is normal. Inject the extra solution, along with the top bubble, back into the vial.
- Withdraw the needle (still needle up), then set the vial down. If you are using a second needle for the injection, swap needles now. Make sure the injection needle is securely fastened (usually a twist-on).
- With the injection needle pointed up, tap the syringe and very slowly squeeze out the final bubbles. You might lose a bit of the solution, but it is important to be patient amount removing all of the significant bubbles (however, you need not worry about the suspended bubbles which are so tiny as to be nearly invisible).
- If you need to change position to make the injection, put the protective cover on over the needle so you can set it down. Some people find it easiest to stand; others prefer to lay on their stomach if the buttock is target. If possible, have someone you trust make the actual injection; it is much easier that way.
- Uncover the needle, grasp the outside of the syringe firmly (finger off the plunger), place the needle against your skin, perpendicular, then bravely push to a depth of roughly 2 1/2 - 3 1/2 cm , (1 - 1 1/3 inches). There should not be much pain past the initial prick. Once the needle is in, try not to shift your weight around or flinch such that the muscles there would move.
- Still holding the outside of the syringe, pull back the plunger to be sure you did not hit a significant blood vessel. If you see no blood in the syringe, then very slowly but firmly depress the plunger. If you do see blood, then withdraw the needle, apply pressure to the site for a minute, then [optionally install a new needle and] try again a few centimeters away, or on the opposite side of your body.
- Remove the needle from your body, replace the protective cover, and dispose of that part into a sharps container, or at least a container of strong composition that cannot be punctured by the used needles.
- It is normal for there to be slight oozing of blood and/or oil from the injection site (and a small bruise later), given the large needle guage. If it oozes for more than a few seconds, apply pressure for a minute. If you are the extraordinarily tidy type, you can also place a dot bandaid over it, but it is not really necessary.
Congratulations!
How Can the Intended Effects of Hormone Therapy Be Maximized and the Dangers Minimised?
- Before starting hormone therapy, take a full physical exam, and have blood drawn to check liver function (enzymes) and clotting factors. If you can possibly afford it, also take tests for thyroid, kidney, electrolyte, lipid (cholesterol), prolactin, sugar, estrogen, and androgen levels. It is also interesting to monitor the skeletal health via the calcium and phosphorus levels, especially if you are more than 40 years old.
- If you take oral estrogen or synthetic progesterone (progestin), repeat the liver and clotting tests a few months after each significant increase of dosage. At the very minimum, recheck them 6 months and 12 months after starting. Even after achieving a stable long-term (> 2yr) oral regimen, it is not a bad idea to recheck the liver and clotting again every couple of years. If you are only on injectible or transdermal hormones, a single recheck 9-12 months after starting should be sufficient, if you are otherwise healthy.
- If you take spironolactone, have an electrolyte test about a month after each significant increase of dosage, especially if you have any known problems with potassium levels.
- Discharge from the nipples may be a sign of a dangerously elevated prolactin level due to intolerance of the estrogen dosage. Immediately take a serum prolactin test. Note that there may be a dramatic spike in the prolactin level, causing significant lactation for up to a week, if a high estrogen dosage is suddenly stopped; this is similar to the process in a female who has just bore her child.
- Be constantly aware of your body so that adjustments can be made if any new problems develop during therapy.
- Have regular medical checkups (minimally every year; more often if you have any significant health problem); pay close attention to vital signs.
- Eat well, and take a good multi-vitamin/mineral supplement to help be sure the body has everything it needs for new development. It might be worth paying special attention to the B vitamens, as it has been reported by some that 1-2mg/day folic acid seems to help increase estrogen assimilation, and that, more generally, B-complex supplements have helped many feel better overall during pre-op level estrogen therapy (just do not go overboard with mega doses). Also, milk thistle has been promoted as a liver tonic, which seems reasonable because it contains silymarin. Silymarin has been shown to protect the liver from free radical damage by directly acting as an antioxidant, as well as by increasing the liver's content of glutathione and superoxide dismutase, which help the liver detoxify a wide range of hormones, drugs, and chemicals. Perhaps most interesting is silymarin's reputed ability to stimulate protein synthesis and the production of new liver cells, suggesting that it may be useful not only in preventing acute liver toxicity, but also in promoting recovery from chronic liver damage.
- Do not start taking the maximum planned dosage of all hormones at once. Start with a low dosage of one, and carefully watch for negative vital signs and symptoms. If there are no problems after 2 months, increase the dosage to the planned level. Wait another 2 months before adding the next hormone or anti-hormone. Do not change the regimen radically or more often than once per 2 months. Give the body time to adjust.
- Use the lowest hormone dosage that affords the desired changes. Not everyone needs the same dosage, because of differences in body weight and genetically-disposed sensitivity to the hormones. Hormone dosage can usually be reduced to a nominal maintenance level after the testes are surgically removed. It is not recommended to take pre-operative dosages of hormones for more than about 3 years.
- Keep your hormone levels as even as you can. If possible, divide oral drugs into twice-daily portions. For injections, if you can stand the hassle and extra cost of syringes, divide your dose for taking every 1-2 weeks rather than letting it go 3-4 weeks.
- Try the daily dosage of a hormone before moving to a sustained-release version, e.g., make sure you do not have adverse allergic or psychological reactions to Provera tablets before you use Depo-Provera (the sustained release intramuscular injection).
- Estrogens delivered orally strain the liver more than other delivery methods. However, it is not highly dangerous unless the liver is already weakened by alcohol, drug use, or infection. It is a good idea to reduce alcohol and other drug intake.
- Susceptibility to hardening of the arteries decreases somewhat, but susceptibility to blood clots, phlebitis (inflammation of lower extremity and pelvic veins), varicose veins, elevated high blood pressure increases somewhat. Stop smoking, reduce stress, and increase aerobic exercise. Investigate severe leg pain by x-ray or ultrasound to determine if it is caused by a blood clot before massaging it. Leg and foot cramping not caused by a blood clot might be reduced with potassium and vitamin E supplements (but one should not take potassium concurrently with spironolactone). Stop or drastically reduce estrogen dosage at least one month before having major surgery that would keep you in bed for more than 1 full day without any walking (to reduce the risk of thrombosis). If you take a significant oral estrogen dosage, consider adding about 80mg/day aspirin to reduce the risk of blood clots; take it with food and liquid to reduce the risk of stomach ulcer--or, better yet, use the enteric safety-coated variety.
- Since spironolactone is a diuretic, anyone taking it should drink plenty of water, especially before and after exercise, and may need to reduce dietary intake of potassium--especially if the kidneys are already stressed.
- Breast cancer risk seem to be low in comparison to females receiving estrogen replacement therapy. Certain studies in females seem to show that the cancer risk is lowered by consistently administering progesterone with the estrogen. Perform monthly breast self-exams, anyway; take mammograms every 2 years before age 40, every year thereafter. Prostate cancer risk is significantly reduced in comparison to males not receiving estrogen therapy. Have the prostate examined once a year if possible, anyway.
Results
A hormone therapy regimen that works well for one person may not for another. If development is not well under way in, say, 6 months, some experimentation may be in order; try different hormone types and/or combinations. However, if you change the regimen very often, it will be difficult to tell which one was working best. Be patient. The obvious exception is if you have a strong adverse effect that you or your physician deem dangerous; in that case you obviously must stop taking the hormone (or anti-hormone) in question. It is unusual for the therapy to not work; the most common cause is the choice of oral preparations since the absorbtion varies among individuals.
Hormone dosage can usually be reduced to a nominal maintenance level after the testes are surgically removed.
How Can One Obtain Hormones?
In the U.S., most reputable therapists and medical doctors who regularly work with transsexuals follow the „Harry Benjamin Standards of Care“ http://www.symposion.com/ijt/ijtc0405.htm, a plan that specifies that one should undergo a minimum of 3 months of psychotherapy to obtain a letter of recommendation to an endocrinologist. One can choose to work with doctors who do not follow the Benjamin Standards, but, in any case, it is a very good idea to meditate and cogitate on the implications for at least 3 months before starting hormone therapy. Some transsexuals find the Benjamin Standards too restrictive--even insulting; others find it worth the trouble to go through the hoop in order to be referred to an endocrinologist who is particularly knowledgeable in the treatment of transsexuals. Choose carefully.
If a sympathetic endocrinologist is not available, try local gynecologists; they are sometimes more understanding, and are used to prescribing estrogens and progesterones.
One should only take hormones that were obtained directly from a licensed pharmaceutical distributor; the quality of drugs obtained through other channels is not only suspect, but likely dangerous--especially those in injectable form.
It is possible to have a health insurance company to cover hormones just like any other prescription drugs, especially if the doctor prescribes them for a "hormone imbalance" or "hormone replacement" rather than "transsexual hormone therapy." When a health insurance company subcontracts out prescription drug coverage to another company, benefits for hormones are not generally questioned since there is little communication between the two companies.
Some people in the U.S. have reportedly taken advantage of the U.S. „FDA Personal Use Import Policy“ http://www.ceri.com/import.htm to purchase hormones directly from international sources. Please note that Confluence Publications does not have any further information about these potential sources.
Are Birth-Control Pills a Good Source of Estrogen?
No. Although early birth-control pills contained significant quantities of estrogen, modern ones do not. A typical birth-control pill now contains a tiny dosage of progesterone, with or without a tiny dosage of estrogen--less than one-tenth the strength required for an effective course of treatment for a pre-op transsexual. If one is absolutely determined to use a particular birth-control pill, then one should carefully study the PDR to understand the dosages of the component hormones of the pill in question, compared to the typical dosages of the same hormones in this FAQ.
Exactly what hormones are available? What Are the Details On Popularity, Dosage, Availability, Contraindications, Adverse Effects, Etc.?
Oestrogens
The following estrogens are popular for treatment of male-to-female transsexuals, and are presented in descending order of preference in the humble opinion of the author:
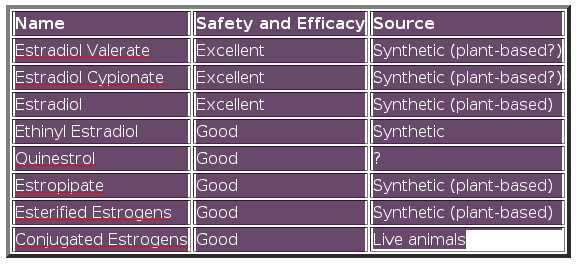
Table 4: Popular estrogens
Estradiol Valerate
Brand Name Manufacturers
Delestrogen by B.M. Squibb
Progynova by Schering
Progynon-Depot by Schering, Germany
Generic Manufacturers
Goldline
Gynogen by Forest
Major
Schein
Steris
Valergen by Hyrex
Pharmacology
Same as endogenous estrogen
Delivery 1, 2mg oral tablets.
Sustained release intramuscular injection.
Typical dosage
Pre-op 20-40mg/3wks injection or 15-30mg/2wks or 7-15mg/1wk injection
Pre-op 8mg/day oral
Post-op 10-20mg/2-3wks injection
Availability
Injection approved by U.S. FDA. Oral tablets may be approved but do not seem to be available in U.S.
Indications
Estrogen replacement therapy in females
Contraindications
Active blood clotting disorders. Estrogen-dependent tumors. History of blood clotting disorders associated with estrogen
use. History of sensitivity to estradiol or any part of the preparation. Known or suspected breast cancer except in appropriately selected
patients.
Adverse reactions
CNS
Convulsions. Dizziness. Headache. Migraine. Mental depression. Spasms of limb and facial muscles.
Eyes
Intolerance to contact lenses. Steepening of corneal curvature.
Gastrointestinal
Abdominal cramps. Bloating. Cholestatic jaundice. Nausea. Vomiting.
Skin
Blotchy skin pigmentation. Localized skin irritation. Loss of scalp hair. Increase of body hair. Red skin patches from capillary congestion.
Other
Blood clotting disorders. Elevated blood pressure. Fluid retention. Glucose intolerance. Increased serum calcium level. Increased sensitivity to light. Liver tumors.
Estradiol Cypionate
Brand Name Manufacturers
Depo-Estradiol by Pharmacia/Upjohn
Generic Manufacturers
Depgen by Hyrex
Dep-gynogen by Forest
Estrocyp by Keene
Goldline
Moore
Rugby
Schein
Steris
Pharmacology
Same as endogenous estrogen
Delivery
Sustained release intramuscular injection, 5mg/ml
Typical dosage
Pre-op 2-4mg/3wks injection or 1.5-3mg/2wks injection??
Post-op 1-2mg/2-3wks injection??
Availability
Approved by U.S. FDA
Indications
Estrogen replacement therapy in females
Contraindications
Active blood clotting disorders. Estrogen-dependent tumors. History of blood clotting disorders associated with estrogen
use. History of sensitivity to estradiol or any part of the preparation. Known or suspected breast cancer except in
appropriately selected patients.
Adverse reactions
CNS
Convulsions. Dizziness. Headache. Migraine. Mental depression. Spasms of limb and facial muscles.
Eyes
Intolerance to contact lenses. Steepening of corneal curvature.
Gastrointestinal
Abdominal cramps. Bloating. Cholestatic jaundice. Nausea. Vomiting.
Skin
Blotchy skin pigmentation. Localized skin irritation. Loss of scalp hair. Increase of body hair. Red skin patches from capillary congestion.
Other
Blood clotting disorders. Elevated blood pressure. Fluid retention. Glucose intolerance. Increased serum calcium
level. Increased sensitivity to light. Liver tumors.
Comments
The reason there are question marks for the pre-op dosage is:
- The author extrapolated from the 1:10 ratio of cypionate:valerate ovarian failure replacement dosages recommended in the 1997 pdr generics
listings, in turn, roughly adjusted against the popular anecdotal valerate pre-op dosage, and;
- The author has found no cypionate anecdotes either to support or change this guess.
Estradiol
Brand Name Manufacturers
Climara by Berlex Labs (film)
Estraderm and Vivelle by Ciba (film)
Estrace by B/M Squibb (oral)
Generic Manufacturers
Apothecon
Geneva
Goldline
Major
Moore
Qualitest
Rugby
Watson
Pharmacology
Same as endogenous estrogen
Delivery
Oral tablets 0.5, 1, 2mg
Extended release film 0.0375, 0.05, 0.075, 0.1mg/24hrs
Typical dosage
Pre-op oral 2-8mg/day, film single 0.1 changed twice weekly
Post-op oral 1-4mg/day, film single 0.05 changed twice weekly
Availability
Approved by U.S. FDA
Indications
Estrogen replacement therapy in females
Contraindications
Active blood clotting disorders. Estrogen-dependent tumors. History of blood clotting disorders associated with estrogen
use. History of sensitivity to estradiol or any part of the preparation. Known or suspected breast cancer except in appropriately selected
patients.
Adverse reactions
CNS
Convulsions. Dizziness. Headache. Migraine. Mental depression. Spasms of limb and facial muscles.
Eyes
Intolerance to contact lenses. Steepening of corneal curvature.
Gastrointestinal
Abdominal cramps. Bloating. Cholestatic jaundice. Nausea. Vomiting.
Skin
Blotchy skin pigmentation. Localized skin irritation. Loss of scalp hair. Increase of body hair. Red skin patches from capillary congestion.
Other
Blood clotting disorders. Elevated blood pressure. Fluid retention. Glucose intolerance. Increased serum calcium level.
Increased sensitivity to light. Liver tumors.
Quinestrol
Brand Name Manufacturers
Estrovis by Parke-Davis
Generic Manufacturers
None
Pharmacology
3-cyclopentylether of ethinyl estradiol. Acts on receptors apparently the same as endogenous estrogen.
Delivery
Oral 0.1mg tablets
Typical dosage
Pre-op ?
Post-op 0.1-0.2mg/wk
Availability
Approved by U.S. FDA
Average Wholesale Price
$141.70/100
Indications
Estrogen replacement therapy in females
Contraindications
Active blood clotting disorders. History of blood clotting disorder in association with estrogen therapy. Known or
suspected breast cancer. Known or suspected estrogen-dependent tumors.
Adverse reactions
CNS
Dizziness. Headache. Mental depression. Migraine. Spasms of limb and facial muscles.
Eyes
Intolerance to contact lenses. Steepening of corneal curvature.
Gastrointestinal
Abdominal cramps. Bloating. Cholestatic jaundice. Nausea. Vomiting.
Skin
Blood eruptions from skin. Blotchy skin pigmentation. Increase of body and facial hair. Loss of scalp hair. Red skin patches from
capillary congestion.
Other
Blood clotting disorders. Breast and liver tumors. Elevated blood pressure. Fluid retention. Gall bladder disease. Increased calcium level
in blood. Increased sensitivity to light. Reduced carbohydrate and glucose tolerance.
Estropipate
Brand Name Manufacturers
Ogen by Pharmacia/Upjohn
Generic Manufacturers
Caremark
Duramed
Goldline
Ortho-est by Ortho Pharm
Qualitest
Rugby
Schein
Pharmacology
Sulfate of estrone, stabilized with piperazine. Apparently acts on receptors the same as endogenous estrogen.
Delivery
Oral 0.75, 1.5, 3mg tablets
Typical dosage
Pre-op ?
Post-op Oral 1.5-9mg/day
Availability
Approved by U.S. FDA
Indications
Estrogen replacement therapy in females
Contraindications
Active blood clotting disorders. Known or suspected breast cancer, unless that is the target. Known or suspected estrogen
dependent tumors.
Adverse reactions
CNS
Dizziness. Headache. Mental depression. Migraine. Spasms of limb and facial muscles.
Eyes
Intolerance to contact lenses. Steepening of corneal curvature.
Gastrointestinal
Abdominal cramps. Bloating. Cholestatic jaundice. Nausea. Vomiting.
Skin
Blood eruptions from skin. Blotchy skin pigmentation. Increase of body and facial hair. Loss of scalp hair. Red skin
patches from capillary congestion.
Other
Blood clotting disorders. Breast tumors. Elevated blood pressure. Fluid retention. Gall bladder disease. Increased
calcium level in blood. Increased sensitivity to light. Reduced carbohydrate tolerance.
Comments
Since estropipate is a „natural estrogenic substance prepared from purified crystalline estrone“, the source is likely to be
pregnant mares, the same as for conjugated and esterified estrogens. Refuting or confirming evidence would be appreciated.
Esterified Oestrogens
Brand Name Manufacturers
Menest by SK Beecham Pharm
Estratab by Solvay
Generic Manufacturers
Cheshire
Pharmacology
Esterified estrogens are a mixture of the sodium salts of the sulfate esters of the estrogenic substances, principally
estrone. They seem to act on estrogenic receptors the same as endogenous estrogen.
Delivery
Oral 0.3, 0.625, 1.25, 2.5mg tablets
Typical dosage
Pre-op 2.5-7.5mg/day
Post-op 1.25mg/day
Availability
Approved by U.S. FDA
Indications
Estrogen replacement therapy in females. Inoperable progressing breast or prostate cancer.
Contraindications
Active blood clotting disorders. Estrogen-dependent tumors. History of blood clotting disorders associated with estrogen
use. History of sensitivity to estradiol or any part of the preparation. Known or suspected breast cancer except in appropriately selected
patients.
Adverse reactions
CNS
Dizziness. Headache. Mental depression. Migraine. Spasms of limb and facial muscles.
Eyes
Intolerance to contact lenses. Steepening of corneal curvature.
Gastrointestinal
Abdominal cramps. Bloating. Cholestatic jaundice. Nausea. Vomiting.
Skin
Blood eruptions from skin. Blotchy skin pigmentation. Increase of body and facial hair. Loss of scalp hair. Red skin
patches from capillary congestion.
Other
Blood clotting disorders. Breast and liver tumors. Elevated blood pressure. Fluid retention. Gall bladder disease. Increased sensitivity
to light. Increased serum calcium level. Reduced glucose tolerance.
Conjugated Oestrogens
Brand Name Manufacturers
Premarin by Wyeth-Ayerst
Generic Manufacturers
None
Pharmacology
Sodium salts of estrogen sulfates. Apparently acts on receptors the same as endogenous estrogen.
Delivery
0.3mg, 0.625, 0.9, 1.25, 2.5mg tablets
Typical dosage
Pre-op Oral 1.25-7.5mg/day
Post-op Oral 0.625-5mg/day
Availability
Approved by U.S. FDA
Indications
Estrogen replacement therapy in females. Treatment of selected breast and prostate cancers.
Contraindications
Active blood clotting disorders. Known or suspected breast cancer, unless that is the intended target. Known or
suspected estrogen dependent tumors.
Adverse reactions
CNS
Dizziness. Headache. Mental depression. Migraine. Spasms of limb and facial muscles.
Eyes
Intolerance of contact lenses. Steepening of corneal curvature.
Gastrointestinal
Abdominal cramps. Bloating. Cholestatic jaundice. Nausea. Vomiting.
Skin
Blood eruptions from skin. Blotchy skin pigmentation. Increase of facial and body hair. Loss of scalp hair. Red skin
patches from capillary congestion.
Other
Blood clotting disorders. Breast tumors. Elevated blood pressure. Fluid retention. Gall bladder disease. Increased
calcium level in blood. Increased sensitivity to light. Reduced carbohydrate tolerance.
Comments
Conjugated estrogens are derived from pregnant mare urine under cruel conditions including continual
confinement, continual standing with no option to lay down or turn around, restriction of drinking water, inadequate
veterinary oversight, killing of the newborn or young foals, then immediate reimpregnation. The pregnancies are repeated
until the mare becomes infertile or sick, at which time she is slaughtered. This treatment has not been directly
witnessed by the author. However, Redwings Horse Sanctuary, World Society for the Protection of Animals, People for the
Ethical Treatment of Animals, and others have researched this issue, interviewed Wyeth-Ayerst representatives, and
directly inspected the farms in question.
Other prescription estrogens are available; however, they are mixed with other drugs, or are intended only for treatment of inoperable cancer, and are therefore not as suitable for treatment of transsexuals.
The following natural sources of phytoestrogens (estrogen-like compounds) have been identified, but the author is not aware of an effective course of treatment using them. They work by weakly binding to estrogen receptors. In males, this may result in a mild feminizing effect (in females, it may give the opposite result, that is, a mild androgenic effect, since the phytoestrogens are competing with endogenous true estrogens for the estrogen receptors). Since phytoestrogens are not nearly as efficacious as true estrogens, huge and potentially toxic amounts of these items would have to be consumed. They are presented in alphabetical order: Black Cohosh (Cimicifuga racemosa), Blue Cohosh, Borrage, Butterfly Weed, Caraway, Chaste Tree or Vitex (Verbenaceae species), Dates, Dill, Dong Quai (Angelica sinensis), False Unicorn root, Fennel seed, Fenugreek, Ginseng, Goats Rue, Gotu Kola, Licorice root, Linseed or Flaxseed, Milk thistle, Motherwort, Pennyroyal (Hedeoma pulegioides), Pleurisy root, Pomegranates, Red Clover Sprouts, Red Raspberry leaf, Southernwood, Soya Flour, Tansy.
Preparations advertized to contain "raw ovaries" from any animal have not been proven to be effective.
Progesterones
The following progesterones are popular for treatment of male-to-female transsexuals and are presented in descending order of preference in the humble opinion of the author:

Progesterone
Brand Name Manufacturers
Utrogestan by Besins-Iscovesco in France
Prometrium by Schering in Canada
Generic Manufacturers
Compound pharmacies advertising unbranded natural progesterone on the web include Bajamar Women's Healthcare Pharmacy and Women's
International Pharmacy.
Pharmacology
Suspension of micronized natural progesterone in oil. Reputedly the same molecule as produced endogenously in females.
Delivery
Custom-packed capsules from a compounding pharmacy.
Vaginal cream.
Dosage
Pre-op 100-400mg/day capsules in conjunction with estrogens.
Post-op 50-400mg/day capsules in conjunction with estrogens.
Unclear how well cream is absorbed.
Availability
Approved by U.S. FDA
Indications
Menopausal discomfort
Contraindications
Active or past blood clotting disorders. Liver dysfunction or disease.
Adverse reactions
Generally mild and transient.
Comments**
Natural progesterone has received rather a lot of attention on women's health support USENET groups such as alt.support.menopause.
Some people call this drug progesterone USP, to differentiate from progestins. USP is an abbreviation for United States Pharmacopeia, a legally recognized compendium of standards for drugs, published by The United States Pharmacopeial Convention, Inc., and revised periodically. It includes assays and tests for the determination of strength, quality, and purity. In other countries the drug is simply referred to as progesterone.
Hydroxyprogesterone Caproate
Brand Name Manufacturers
Caposten by ?
Capton by ?
Caprosteron by ?
Hormofort by ?
Delalutin by ?
Depolut by ?
Estralutin by ?
Neolutin by ?
Primolut-Depot by ?
Progesteron-retard by ?
Prolutin-Depot by ?
Syngynon by ?
Generic Manufacturers
Hylutin by Hyrex
Moore, H.L.
Rugby
Schein
Steris
Pharmacology
Progestogen (progesterone derivative)
Delivery
250mg/ml sustained-release intramuscular injection
Typical dosage
125mg/2wks intramuscular injection
Availability
Approved by U.S. FDA
Indications
Unusual menstrual bleeding. Endometriosis
Contraindications
Active or past blood clotting disorders. Cerebral clotting or haemorrhage.
Adverse reactions
CNS
Headache. Insomnia. Loss of coordination. Mental depression. Sleepiness. Slurred speech. Weakness, numbness, or pain in extremeties.
Eyes
Change of vision.
Gastrointestinal
Cholestatic jaundice. Nausea.
Skin
Skin discoloration, rash, itching, and other allergic reactions.
Other
Blood clotting disorders. Chest pain. Decreased glucose tolerance. Fever. Fluid retention. Shortness of breath.
Medroxyprogesterone Acetate
Brand Name Manufacturers
Amen by Carnrick
Curretab by Solvay
Provera and Depo-Provera by Pharmacia/Upjohn
Generic Manufacturers
Cycrin by Esi Lederle Generics
Geneva
Goldline
Greenstone
Intl Labs
Major
Martec
Moore
Parmed
PD-RX
Qualitest
RID
Rosemont
Rugby
Schein
Pharmacology
Progestin (progesterone derivative)
Delivery
2.5, 5, 10mg tablets
400mg/ml sustained-release intramuscular injection (brand-name only)
Typical dosage
Pre-op 2.5-10mg/day tablets in conjunction with estrogens
Pre-op 50mg/2weeks injectible in conjunction with estrogens
Pre-op ? for neutering without estrogens
Post-op ?
Availability
Approved by U.S. FDA
Indications
Endometrial and kidney cancer. Unusual menstrual bleeding.
Contraindications
Active or past blood clotting disorders. Known or suspected breast or gonadal tumors. Known sensitivity to
medroxyprogesterone acetate. Liver dysfunction or disease.
Adverse reactions
CNS
Headache. Insomnia. Loss of coordination. Mental depression. Sleepiness. Slurred speech. Weakness, numbness, or pain
in extremeties.
Eyes
Change of vision.
Gastrointestinal
Cholestatic jaundice. Nausea.
Skin
Skin discoloration, rash, itching, and other allergic reactions.
Other
Blood clotting disorders. Chest pain. Decreased glucose tolerance. Fever. Fluid retention. Shortness of breath.
Comments
There are many anecdotal reports of inexplicable or exacerbated depression while taking this drug. In that case, natural progesterone is
indicated.
Upjohn claims that the bioavailability of Provera is higher than generic formulations.
The article "Gender Dysphoria Update" by Blaine R. Beemer (originally published in Journal of Psychosocial Nursing and Mental Health Services, 1996: 34(4), 12-19) reports that clients at Vancouver (BC) "routine receive the progestin medroxyprogesterone acetate (Provera)" and asserts that apart „from its effect as an antiandrogen, medroxyprogesterone has been shown to promote bone formation, and may counter the bone loss that might occur with the blockade of male hormones,“ citing as a reference: Prior, JC, Vigna, YM, Barr, SI, Rexworthy, C, & Lentle, BC (1994), „Cyclic medroxyprogesterone treatment increases bone density: A controlled trial in active women with menstrual cycle disturbances.“ American Journal of Medicine, 96, 521-530. A question to consider: does the medroxyprogesterone administration have to be cyclic to have the bone density effect?
Norethindrone Acetate
Brand Name Manufacturers
None
Generic Manufacturers
Aygestin by Esi Lederle Generics
Pharmacology
Progestin
Delivery
Oral 5mg tablets
Typical dosage
Pre-op 2.5-15mg/day
Post-op ?
Availability
Approved by U.S. FDA
Indications
Endometriosis. Unusual menstrual bleeding.
Contraindications
Blood clotting disorders. Known or suspected breast or gonadal cancer. Known sensitivity to norethindrone acetate. Liver
dysfunction or disease.
Adverse reactions
CNS
Insomnia. Mental depression. Sleepiness.
Eyes
foo.
Gastrointestinal
Cholestatic jaundice. Nausea.
Skin
Acne. Increase of body and facial hair. Loss of scalp hair.
Other
Blood clotting disorders. Fever. Fluid retention. Mild to severe allergic reactions.
The following natural sources of phytoprogesterones (progesterone-like compounds) have been identified, but the author is not aware of an effective course of treatment using them. Since phytoprogesterones are not nearly as efficacious as true progesterones, huge and potentially toxic amounts of these unrefined items would have to be consumed. They are presented in alphabetical order: Suma, Vitex, Wild or Mexican Yam.
Anti-androgens
The following anti-androgens are popular for treatment of pre-operative male-to-female transsexuals. They are presented in descending order of preference in the humble opinion of the author:
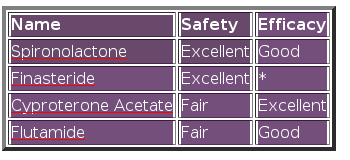
Table 6: Popular anti-androgens
Spironolactone
Brand Name Manufacturers
Aldactone by Searle
Generic Manufacturers
Caremark
Cheshire
Geneva
Goldline
Heartland
Major
Moore, H.L.
Mutual
Mylan
Parmed
PD-RX
Purepac
Qualitest
Raway
Rugby
UDL
URL
Vanguard
Pharmacology
Weak androgen receptor antagonist
Delivery
25, 50, 100mg oral tablets
Typical dosage
Pre-op 100-300mg/day
Post-op 50mg/day
Availability
Approved by U.S. FDA
Indications
Congestive heart failure. Elevated blood pressure. Fluid retention. Hyperaldasteronism. Inadequate potassium retention. Liver cirrhosis.
Contraindications
Elevated potassium levels. Inadequate urine production. Kidney disfunction.
Adverse reactions
CNS
Confusion. Dizziness. Drowsiness. Headache. Lethargy. Loss of precise motor control.
Gastrointestinal
Cramping. Diarrhea. Dry mouth. Gastric ulceration and other stomach inflammation. Vomiting.
Skin
Acne. Itchy, fluid-filled patches of skin. Increase of body and facial hair. Red skin patches from capillary congestion.
Other
Deepening of the voice. Drug fever. Pottasium retention. Severe decrease of blood granulocytes. Sodium loss.
Comments
One person reported suicidal depression as an adverse effect. The brand-name formulation tastes awful; the generic
formulation is much less offensive.
Finasteride
Brand Name Manufacturers
Proscar by Merck
Propecia by Merck
Generic Manufacturers
None
Pharmacology
Androgen conversion inhibitor. Inhibits the production of dihydrotestosterone (DHT) from testosterone by inhibiting the
binding of 5a-reductase, which is the enzyme responsible for converting testosterone to DHT. DHT is the active androgen
found in the skin and prostate gland, and is associated with the development of male pattern baldness, excess body hair,
and benign prostatic hypertrophy. Not suitable as a general anti-androgen since it only affects DHT production. However,
it seems to be more helpful in counteracting male-pattern baldness and excess body hair than general anti-androgens.
Delivery
5mg oral tablets (Proscar)
1mg oral tablets (Propecia)
Typical dosage
Pre-op 0.05-1mg/day
Post-op 0.05-1mg/day
(See comments below)
Availability
Approved by U.S. FDA
Indications
Benign prostate enlargement
Contraindications
Hypersensitivity to any component of the product.
Adverse reactions
Generally mild and transient
Comments
Anecdotal evidence strongly suggests that pill fragments
taken daily are just as effective as taking the entire pill. There might
issues with oxidation, so avoid handling the unused fragments, and keep
them in a small, air-tight container.
Do not let a female who is pregnant, or might be pregnant, anywhere near finasteride fragments or powder. It is a strong teratogen, known to cause genital deformity in the male fetus.
Based on rabbit and rat studies, there may be a slight effect on male fertility that would reverse within 6 weeks of discontinuing.
Cyproterone Acetate
Brand Name Manufacturers
Androcur by Schering AG, Farma (Germany)
Cyproteron by NM Pharma (England)
Generic Manufacturers
?
Pharmacology
Androgen receptor antagonist. Weak gonadal androgen production inhibitor. Weak progesterone.
Delivery
10mg, 50mg oral tablets
Typical dosage
Pre-op 10mg/wk-100mg/day (See comment below)
Post-op not recommended
Availability
Not approved by U.S. FDA
Indications
Acne and/or overactive oil glands. Androgen dependent loss of scalp hair. Hirsutism. Inoperable prostate tumors.
Contraindications
Lactation. Dubin-Johnson syndrome. Liver disease or tumor. Previous or existing blood clotting disorder. Rotor
syndrome. Severe chronic-depression. Severe diabetes with vascular changes. Sickle-cell anaemia. Wasting diseases
(with the exception of prostate tumor).
Adverse reactions
CNS
Headache. Lessened ability to concentrate. Mental depression. Tiredness.
Gastrointestinal
Nausea.
Other
Blood clotting disorders. Carbohydrate metabolism changes. Liver dysfunction or tumors. Shortness of breath.
Comments
The extreme range of dosage comes from input that some
people find 10mg/wk sufficient to induce total impotence, and yet others
take as much as 200mg/day with no obvious short-term adverse effects.
Given this range, it would seem prudent to start on the low side and
work your way up only if necessary. More than 100mg/day is generally
considered excessive.
Flutamide
Brand Name Manufacturers
Eulexin by Schering
Generic Manufacturers
None
Pharmacology
Strong androgen receptor antagonist
Delivery
125mg oral capsules
Typical dosage
Pre-op 375-750mg/daily in 3 divided doses
Availability
Approved by U.S. FDA
Indications
Androgen-sensitive prostate cancer
Contraindications
Hypersensitivity to any component of the product
Adverse reactions
CNS
Anxiety. Confusion. Depression. Drowsiness. Nervousness.
Gastrointestinal
Anorexia. Diarrhea. Nausea. Vomiting.
Other
Decreased leukocyte blood cell count. Decreased platelet blood cell count. Decreased red blood cell count. Fluid retention. Elevated blood
pressure. Hot flashes. Liver disease.
Comments Although it is not a general anti-androgen, finasteride coadministered with estrogen is very helpful to halt--and in some cases, partly reverse--male-pattern baldness. Some people report that it also helps to reduce excess body hair.
Cyproterone acetate is a very strong anti-androgen but also causes strong adverse effects in some people.
Prescription adrenal androgen production inhibitors are available but not listed because adrenal androgen production is insignificant (i.e., about the same as in females) in comparison to gonadal adrenal production. Adrenal androgens are best ignored, or if absolutely necessary, countered with finasteride.
Other prescription anti-androgens are available but not listed because their primary indication is not as an anti-androgen, and/or because the adverse effects are dangerous when weighed against the possible benefit.
The following natural sources of phytoantiandrogens (anti-androgen-like compounds) have been identified, but the author is not aware of an effective course of treatment using them. Since phytoantiandrogens are not nearly as efficacious as true antiandrogens, huge and potentially toxic amounts of these items would have to be consumed. They are presented in alphabetical order: Saw Palmetto.
Other Anti-Hormones (GnRH Agonists)
These pharmaceuticals can be used to dramatically reduce gonadal hormone production in both males and females. They are used mainly by pediatricians to reduce precocious puberty, so it might be difficult to persuade a doctor to prescribe them for an adult. Also, they are very expensive. None the less, this type of chemical castration is worth investigating for those cases when the pre-operative male-to-female cannot take the hormones of choice because of other health problems (e.g., hormone dependent tumors or blood clotting disorders), and cannot yet have the surgery performed (note that such a problem is quite rare). They are presented in descending order of preference in the humble opinion of the author:

Table 7: Anti-hormones
Goserelin Acetate
Brand Name Manufacturers
Zoladex by Zeneca
Generic Manufacturers
None
Pharmacology
GnRH agonist. After an initial stimulating phase, the pituitary is desensitized to GnRH, which causes it to stop producing LH,
which in turn dramatically decreases gonadal production of hormones within a month.
Delivery
Sustained release subcutaneous injection 3.6, 10.8mg
Typical dosage
Pre-op 3.6mg/month
(3.6mg implant is for 1 month; 10.8 mg implant is for 3 months)
Availability
Approved by U.S. FDA
Indications
Androgen-sensitive prostate cancer
Contraindications
Known hypersensitivity to GnRH, GnRH analogues, or any of the components of the product
Adverse reactions
CNS
Dizziness. Insomnia. Lethargy.
Gastrointestinal
Anorexia. Nausea.
Skin
Sweating.
Other
Congestive heart failure. Fluid retention. Hot flashes. Increased calcium level in blood. Mild to severe allergic reactions. Obstructive
pulmonary disease. Ureteral and spinal compression.
Nafarelin Acetate
Brand Name Manufacturers
Synarel by Searle
Generic Manufacturers
None
Pharmacology
GnRH agonist. After an initial stimulating phase, The pituitary is desensitized to GnRH, which causes it to stop producing LH,
which in turn dramatically decreases gonadal production of hormones within one month.
Delivery
Nasal spray
Typical dosage
Pre-op 1600mcg/day (2 sprays into each nostril twice a day)
Availability
Approved by U.S. FDA
Indications
Central precocious puberty. Endometriosis.
Contraindications
Hypersensitivity to GnRH, GnRH agonists analogs or any component of the product.
Adverse reactions
CNS
Headache. Insomnia. Mental depression.
Skin
Acne. Body odor. Increase of body and facial hair. Itchiness. Itchy, fluid-filled patches of skin. Oily skin. Rash. Vaginal dryness.
Other
Chest pain. Fluid retention. Hot flashes. Muscle pain. Nasal irritation. Ovarian cysts. Shortness of breath. Vaginal bleeding.
Leuprolide Acetate
Brand Name Manufacturers
Lupron by Tap
Generic Manufacturers
None
Pharmacology
GnRH agonist. After an initial stimulating phase, the pituitary is desensitized to GnRH, which causes it to stop producing LH,
which in turn dramatically decreases gonadal production of hormones within one month.
Delivery
5, 7.5, 11.25, 15, 22.5mg sustained-release intramuscular injection
Typical dosage
Pre-op 3.75-7.5mg/month
Availability
Approved by U.S. FDA
Indications
Advanced prostate cancer. Endometriosis.
Contraindications
Hypersensitivity to GnRH or GnRH analogs.
Adverse reactions
CNS
Anxiety. Delusions. Dizziness. Headache. Hearing disorders. Insomnia. Memory disorder. Nerve disorders. Personality disorder.
Eyes
Eye disorders.
Gastrointestinal
Anorexia. Constipation. Coughing up blood. Dry mouth. Nausea. Thirst. Vomiting.
Skin
Change of facial and body hair. Skin rash.
Other
Ankylosing spondylosis. Blood in the urine. Bone and muscle pain.
Change in heart electrical activity. Congestive heart failure. Decrease
of bone density. Decreased tolerance of protein. Decreased red blood
cell count. Decreased white blood cell count. Difficulty urinating.
Elevated blood pressure. Elevated LDH. Elevated phosphorus. Escape of
blood into the tissues from ruptured blood vessels. Fluid retention.
Hair loss. Hot flashes. Increased heart beat rate. Increased uric acid.
Increased urination frequency or urgency. Lactation. Liver disorder.
Loss of strength. Low blood pressure. Lymphadenopathy. Mild to extreme
allergic reaction. Palpitations. Pelvic fibrosis. Penile swelling.
Prostate pain. Pulmonary disorders. Respiratory disorders. Temporary
increase of hormone production. Temporary suspension of respiration and
circulation.
Where is there more information about hormones?
Medline Articles
These are the articles in Medline containing the words "transsexual" and "hormone" and less than 10 years old, as of 1998 May, courtesy of the U.S. National Institute of Health.
35 citations found
Arch Sex Behav 1998 Apr; 27(2): 209-226
Endocrine therapy of transsexualism and potential complications of long-term treatment
Futterweit W
Division of Endocrinology, Mount Sinai School of Medicine, New York, New York 10029, USA.
[Medline record in process]
Physiological principles of the interrelationship of sex hormones and their regulation are the foundation of understanding appropriate treatment of the transsexual patient. While both genetic males and females have estrogens and androgens, the quantitative sex hormone production is genetically predetermined by sex hormone production both in the gonads and via peripheral conversion of hormone precursors to sex steroids. Sex hormones exert a negative feedback on the hypothalamus and pituitary gland whereby gonadotropin-releasing hormone (GnRH), pituitary luteinizing hormone (LH), and follicle-stimulating hormone (FSH) are regulated or suppressed by the endogenous levels of these hormones. Sex hormonal therapy induces attenuated GnRH stimulation of LH and FSH causing a reduction of serum sex hormone levels. It is clear that estrogen as well as androgen therapy have a dual role: (i) induction of feminization or virilization and (ii) suppression of the hypothalamic-pituitary-gonadal axis leading to a reduction of endogenous estradiol or testosterone secretion. Cross-sex hormonal treatment may have substantial medical side effects. The smallest dosage of hormonal therapy compatible with the above clinical aims should be used.
PMID: 9562902, UI: 98223997
J Clin Endocrinol Metab 1998 Feb; 83(2): 550-553
Effects of sex steroids on plasma total homocysteine levels: a study in transsexual males and females
Giltay EJ, Hoogeveen EK, Elbers JM, Gooren LJ, Asscheman H, Stehouwer CD
Department of Endocrinology, Division of Andrology, Hospital Vrije Universiteit, Amsterdam, The Netherlands.
Plasma total homocysteine (tHcy) levels are higher in men vs. premenopausal women, but it is not known whether this difference is related to sex steroids. The effects of cross-sex hormone administration on plasma tHcy levels were therefore investigated. Plasma tHcy levels were measured at baseline and after 4 months of treatment in 17 male-to-female (M-->F) transsexuals treated with ethinyl estradiol (100 micrograms/day), in combination with the antiandrogen, cyproterone acetate (100 mg/day), and in 17 female-to-male (F-->M) transsexuals treated with testosterone esters (250 mg/2 weeks, im). In M-->F transsexuals, the plasma tHcy level decreased from geometric mean 8.2 mumol/L to 5.7 mumol/L (P < 0.001); and in F-->M transsexuals, it increased from 7.7 mumol/L to 9.0 mumol/L (P = 0.005). In M-->F transsexuals, changes in serum sex hormone-binding globulin levels correlated negatively, and changes in plasma creatinine and albumin levels correlated positively, with changes in plasma tHcy levels. In F-->M transsexuals, changes in serum 17 beta-estradiol levels correlated negatively, and changes in plasma creatinine levels correlated positively, with changes in plasma tHcy levels. We conclude that tHcy levels decrease after estrogen + antiandrogen administration to male (transsexual) subjects, and levels increase after androgen administration to female (transsexual) subjects. These changes may be both primary and secondary to the anabolic/catabolic effects, as reflected by changes of creatinine and albumin levels after cross-sex hormone administration.
PMID: 9467573, UI: 98128702
Arch Sex Behav 1997 Dec; 26(6): 589-605
Transvestism: a survey of 1032 cross-dressers
Docter RF, Prince V
Department of Psychology, California State University, Northridge 91330-8255, USA.
One thousand and thirty-two male periodic cross-dressers (transvestites) responded to an anonymous survey patterned after Prince and Bentler's (1972) report. With few exceptions, the findings are closely related to the 1972 survey results. Eighty-seven percent described themselves as heterosexual. All except 17% had married and 60% were married at the time of this survey. Topics surveyed included demographic, childhood, and family variables, sexual orientation and sexual behavior, cross-gender identity, cross-gender role behavior, future plans to live entirely as a woman, and utilization of counseling or mental health services. Of the present sample, 45% reported seeking counseling compared to 24% of the 1972 survey, and those reporting strong transsexual inclinations were up by 5%. Today's transvestites strongly prefer both their masculine and feminine selves equally. A second research objective was to identify variables discriminating between so-called Nuclear (stable, periodic cross-dressers) and Marginal transvestites (more transgendered or transsexually inclined); 10 strongly discriminating parameters were found. The most important are (i) cross-gender identity, (ii) commitment to live entirely as a woman, (iii) taking steps toward body feminization, (iv) low sexual arousal to cross-dressing. Neither age nor experience as a cross-dresser were found to be correlates of cross-gender identity. Although the present generation of transvestites describe themselves much as did similar subjects 20 years ago, the percentage migrating toward full-time living as a woman is greater.
PMID: 9415796, UI: 98077637
Clin Endocrinol (Oxf) 1997 Sep; 47(3): 337-342
Mortality and morbidity in transsexual subjects treated with cross-sex hormones
van Kesteren PJ, Asscheman H, Megens JA, Gooren LJ
Department of Andrology, Free University Hospital, Amsterdam, The Netherlands.
OBJECTIVE: The optimum steroid hormone treatment regimes for transsexual subjects has not yet been established. We have investigated the mortality and morbidity figures in a large group of transsexual subjects receiving cross-sex hormone treatment. DESIGN: A retrospective, descriptive study in a university teaching hospital. SUBJECTS: Eight hundred and sixteen male-to-female (M-->F) and 293 female-to-male (F-->M) transsexuals. INTERVENTIONS: Subjects had been treated with cross-sex hormones for a total of 10,152 patient-years. OUTCOME MEASURES: Standardized mortality and incidence ratios were calculated from the general Dutch population (age- and gender-adjusted) and were also compared to side effects of cross-sex hormones in transsexuals reported in the literature. RESULTS: In both the M-->F and F-->M transsexuals, total mortality was not higher than in the general population and, largely, the observed mortality could not be related to hormone treatment. Venous thromboembolism was the major complication in M-->F transsexuals treated with oral oestrogens and anti-androgens, but fewer cases were observed since the introduction of transdermal oestradiol in the treatment of transsexuals over 40 years of age. No cases of breast carcinoma but one case of prostatic carcinoma were encountered in our population. No serious morbidity was observed which could be related to androgen treatment in the F-->M transsexuals. CONCLUSION: Mortality in male-to-female and female-to-male transsexuals is not increased during cross-sex hormone treatment. Transdermal oestradiol administration is recommended in male-to-female transsexuals, particularly in the population over 40 years in whom a high incidence of venous thromboembolism was observed with oral oestrogens. It seems that in view of the deep psychological needs of transsexuals to undergo sex reassignment, our treatment schedule of cross-sex hormone administration is acceptably safe.
PMID: 9373456, UI: 98040787
Arch Sex Behav 1997 Apr; 26(2): 143-157
Anthropometrical measurements and androgen levels in males, females, and hormonally untreated female-to-male transsexuals
Bosinski HA, Schroder I, Peter M, Arndt R, Wille R, Sippell WG
Department of Sexual Medicine, Medical School, Christian Albrechts University Keil, Germany.
To elucidate the relationship between body build, androgens, and transsexual gender identity, anthropometric measurements were assessed in 15 hormonally untreated female-to-male-transsexuals (FMT). Nineteen healthy women (CF) (X = 22 years; 2 months), and 21 healthy men (CM) (X = 23; 7) were enrolled as controls. Baseline levels of testosterone (T; ng/dl), androstenedione (A4; ng/dl), dehydroepiandrosterone sulfate (DHEAS; ng/ml), and sex-hormone binding globulin (SHBG; microgram/ml) were assessed in 12 FMT, 15 CF, and in all CM. No control was under hormonal medication (including contraceptives). Absolute measurements in FMT were in accordance with their biological sex: they showed only small differences from the CF. However, FMT differed from CF in 7 of 14 sex-dimorphic indices of masculinity/femininity in body build. Of these 14 indices, 9 did not show a difference between FMT and CM. Hence, FMT presented a more masculine body build, particularly in fat distribution and bone proportions. Levels of T and A4 were significantly higher in FMT than in CF (T: 54.0 +/- 13.8 vs. 41.1 +/- 12.8; A4: 244.8 +/- 73.0 vs. 190.5 +/- 49.3), while DHEAS was higher in CM (3335 +/- 951) than in CF (2333 +/- 793) and in FMT (2679 +/- 1089). Altogether, 83.3% of FMT and 33.3% of CF were above normal values for at least one measured androgen. SHBG in FMT (1.21 +/- 0.70) and CF (1.87 +/- 0.91) was higher than in CM (0.49 +/- 0.18) and tended to be higher in CF than in FMT. Unbound T (T/SHBG ratio) was higher in FMT (72.0 +/- 67.6) than in CF (26.4 +/- 15.1) and correlated positively with manly body shape. Findings are discussed in relation to etiology of transsexualism.
PMID: 9101031, UI: 97255684
J Am Acad Child Adolesc Psychiatry 1997 Feb; 36(2): 263-271
Sex reassignment of adolescent transsexuals: a follow-up study
Cohen-Kettenis PT, van Goozen SH
Department of Child and Adolescent Psychiatry, Rudolph Magnus Institute of Neurosciences, Utrecht, The Netherlands.
OBJECTIVE: To investigate postoperative functioning of the first 22 consecutive adolescent transsexual patients of our gender clinic who underwent sex reassignment surgery. METHOD: The subjects were interviewed by an independent psychologist and filled out a test battery containing questionnaires on their psychological, social, and sexual functioning. All subjects had undergone surgery no less than 1 year before the study took place. Twelve subjects had started hormone treatment between 16 and 18 years of age. The posttreatment data of each patient were compared with his or her own pretreatment data. RESULTS: Postoperatively the group was no longer gender-dysphoric; they scored in the normal range with respect to a number of different psychological measures and they were socially functioning quite well. Not a single subject expressed feelings of regret concerning the decision to undergo sex reassignment. CONCLUSIONS: Starting the sex reassignment procedure before adulthood results in favorable postoperative functioning, provided that careful diagnosis takes place in a specialized gender team and that the criteria for starting the procedure early are stringent.
PMID: 9031580, UI: 97183473
Contracept Fertil Sex 1996 Nov; 24(11): 834-846
Role of hormones in sexual dysfunctions, homosexuality, transsexualism and deviant sexual behavior: diagnostic and therapeutic consequences
[Article in French]
Buvat J, Lemaire A, Ratajczyk J
Association pour l'etude de la pathologie de l'appareil reproducteur et de la psychosomatique (EPARP), Lille.
Hormones only play a minor role in sexual dysfunctions. They are clearly involved only in erectile dysfunction. Total testosterone is low in 8% of those patients, but only 32% of them are improved with androgen therapy. Free testosterone is also electively decreased in 30% and bioavailable (non SHBG bound) testosterone in 15%. However androgen-therapy is still less effective in these subgroups. Plain hyperprolactinemia is found in only 0,7% of the cases. Half of them result from a pituitary adenoma. The other endocrine dysfunctions are still scarcer. This data cannot justify a systematic determination of serum prolactin and testosterone in sexual dysfunctions. A cost effective hormonal screening is proposed, whereas the role of androgen-therapy in erectile dysfunction with or without hypogonadism is discussed. The hypothesis of an "inverted brain sexual differentiation" in homosexuality and for transsexualism, resulting from an abnormal antenatal endocrine milieu is reviewed. It cannot obviously explain by itself these conditions, but some amazing morphological findings in transsexual people do not permit to totally refute it. Lastly the role of androgens in paraphilia and parapaphilia related disorders seems limited to the arousal of an abnormal sexual behaviour previously scheduled by non hormonal mechanisms. However anti-androgens are in such cases one of the main effective treatment.
Publication Types:
- Review
- Review, tutorial
PMID: 8991588, UI: 97114880
Exp Clin Endocrinol Diabetes 1996; 104(6): 413-419
Multistep treatment concept of transsexual patients
Schlatterer K, von Werder K, Stalla GK
Max-Planck-Institute of Psychiatry, Dept. Endocrinology, Munchen, Germany.
Here we present a pragmatic multistep approach for the treatment of transsexual patients. The importance of an individually designed cross-gender hormone replacement therapy embedded in a multidisciplinary treatment concept, provided by psychiatrists, endocrinologists and surgeons, is demonstrated. Following this concept outcome of therapy has been improved in the last years. Over the last 5 years we have gained substantial experience in the cross-gender hormone treatment of transsexual patients. By continuous follow-up examinations and therapy adjustment the risk of side effects accompanying this therapy has been significantly minimized. This report is designed as a guideline to the clinical endocrinologist for the handling and treatment of transsexual patients.
Publication Types:
- Review
- Review, tutorial
PMID: 9021341, UI: 97173435
Clin Endocrinol (Oxf) 1995 Oct; 43(4): 415-421
Effects of gonadal androgens and oestrogens on adrenal androgen levels
Polderman KH, Gooren LJ, van der Veen EA
Department of Internal Medicine, Free University Hospital, Amsterdam, The Netherlands.
OBJECTIVE: The physiological role of adrenal androgens in humans remains unclear. Furthermore, there are few data on the relation between sex steroids and adrenal androgen production. We have assessed the effects of sex steroid hormone administration on adrenal androgen levels, by studying a large group of transsexual patients. DESIGN: A non-randomized intervention. SETTING: A university teaching hospital. PATIENTS: Thirty-one male-to-female and 22 female-to-male transsexual patients. MEASUREMENTS: Plasma levels of adrenal androgens were measured in a group of male-to-female and female-to-male transsexual patients both before and during cross-gender hormone treatment. This treatment involves administration of testosterone esters to women and of ethinyloestradiol and cyproterone acetate to men. RESULTS: High dose sex steroid administration had marked effects on adrenal androgens levels, which decreased by 27-48% in males treated with ethinyloestradiol and increased by 23-70% in females treated with testosterone. CONCLUSION: We conclude that administration of high doses of testosterone and oestradiol exert opposing effects on adrenal androgen production.
Publication Types:
- Clinical trial
Comments:
- Comment in: Clin Endocrinol (Oxf) 1996 Apr;44(4):490-1
PMID: 7586614, UI: 96046830
Arch Androl 1995 Sep; 35(2): 127-133
Aromatization of [4-14C]testosterone to [14C]estradiol-17 beta by testicular tissue from male-to-female transsexuals on estrogen therapy
Gunasegaram R, Loganath A, Peh KL, Ratnam SS
Institute of Science and Forensic Medicine, National Blood Centre, Singapore, Republic of Singapore.
Abnormality in testicular biosynthesis of sex hormones may accompany or cause transsexualism. With a view to determine whether the cells of male transsexual testicular tissue possess aromatic cytochrome P-450 activity, homogenates of the tissue from 4 phenotypic male-to-female transsexuals (2 Chinese and 2 Malays, aged 23-34 years) with XY karyotype who had been on estrogen therapy for 9-17 years until orchidectomy were incubated with [4-14C]testosterone as substrate. Using reverse-isotope dilution analysis, identity was established for [14C]estradiol-17 beta. No such metabolite was found in parallel incubations of heat-denatured enzymes. The extent of enzyme-catalyzed conversion was 9.3 x 10(-3) to 3.4 x 10(-2)%. The specific biosynthetic aromatase cytochrome P-450 activity contained in the tissue system can synthesize [14C]estradiol-17 beta from [4-14C]testosterone. Elevated aromatic expression in situ may have important effects on target tissues.
PMID: 8579473, UI: 96103792
Psyche (Stuttg) 1995 Sep; 49(9-10): 811-837
Transsexual wish and and cis-sexual defense
[Article in German]
Sigusch V
Klinikum, J.W. Goethe-Universitat, Abteilung fur Sexualwissenschaft, Frankfurt am Main.
The desire of trans-sexual patients to appropriate the physical attributes of the opposite sex via hormone treatment or surgery confronts psychoanalysis and sex research with problems that are not only psychological in nature. Sigusch uses the term "defence" here not only in a technical sense but also in an epistemological, discourse-analytic and disposition-analytic register. The general dispositions determining the reality of gender and gender difference are "cis-sexual" in nature. The polarity they display extends to the physical and manifests itself in the form of sexual binarism. The author coins the consciously neologistic term "cis-sexual" to describe individuals where physical gender and gender identity are completely and "naturally" co-extensive. Only the realization that "second-nature" is in reality primary can trigger the detotalization of so-called trans-sexuality that Sigusch is concerned to call into question.
Publication Types:
- Review
- Review, tutorial
PMID: 7480808, UI: 96068432
J Endocrinol Invest 1995 Jun; 18(6): 420-426
The effect of non-steroidal antiandrogen flutamide on luteinizing hormone pulsatile secretion in male-to-female transsexual subjects
Giusti M, Falivene MR, Carraro A, Cuttica CM, Valenti S, Giordano G
Cattedra di Endocrinologia, University of Genova, Italy.
We evaluated LH pulsatile patterns before and 4 weeks after the oral administration of flutamide (750 mg/day) in 9 male-to-female transsexuals (age range 17-28 yr) requesting gender reassignment. Flutamide was given to explore the feedback role of androgens on the LHRH-LH unit in LH pulsatility in transsexuals. Seven normal age-matched men served as a control group, without receiving flutamide, due to ethical considerations. LH pulsatility was evaluated on samples collected every 15 min for 360 min. FSH, PRL, cortisol, SHBG and sex steroids were evaluated on pooled samples. LH pulses were analyzed by the Santen and Bardin algorithm, slightly modified. No differences in FSH, PRL, total- or free-testosterone, estradiol and SHBG levels were noted between transsexuals and controls. Normal circadian cortisol decline was observed in all subjects. Mean LH levels (p < 0.05) and LH pulses (p < 0.01) were significantly lower in transsexuals. Flutamide induced an increase in mean LH and testosterone levels (p < 0.01). After flutamide administration there was an increase in LH pulse frequency (P < 0.01) and the frequency and amplitude of LH pulses in transsexuals were restored to levels observed in controls. No differences in FSH, PRL or estradiol levels were found after flutamide. These data suggest that a decrease in LH pulse frequency could be an endocrine marker in male-to-female transsexuals. An increase in endogenous androgen negative feed-back could be speculated in these subjects. However, normal testosterone levels indirectly suggest that a normal that a normal qualitative LH secretion is maintained.
Publication Types:
- Clinical trial
PMID: 7594235, UI: 96037312
Int J Androl 1995 Apr; 18(2): 88-96
Quantification of androgen receptor and follicle-stimulating hormone receptor mRNA levels in human and monkey testes by a ribonuclease-protection assay
Dankbar B, Sohn M, Nieschlag E, Gromoll J
Institute of Reproductive Medicine, University of Munster, Germany.
A sensitive, solution-hybridization ribonuclease-protection assay (RPA) was established to quantify the expression of mRNA for the androgen receptor (AR) and follicle-stimulating hormone receptor (FSHR) in total RNA samples isolated from tissues of the cynomolgous monkey, human testes obtained from elderly patients undergoing orchidectomy because of prostatic carcinoma or from transsexual men undergoing gender reassignment as well as human cell lines DU 145, REP and RVP. Sensitivity experiments revealed that, in the human and monkey, 1-2 micrograms of total RNA were sufficient to achieve quantifiable signals of the different receptor mRNA species. Quantification of AR and FSHR mRNA levels showed a 1.7-fold higher expression of AR mRNA and a 2.4-fold higher expression of FSHR mRNA in the monkey testes compared to human testes from patients with prostatic carcinoma. Normal spermatogenesis in both human and monkey testes indicated no relationship between spermatogenic status and receptor expression. The significantly lower expression of AR and FSHR mRNA in humans than in monkeys might therefore be either age- or species-related. Quantification of mRNA for AR and FSHR in the testis of the transsexual patients undergoing oestrogen and antiandrogen treatment displayed a drastic increase (4.5-fold) in mRNA for the AR, whereas mRNA for the FSHR was barely detectable. Due to its high sensitivity, reproducibility and its ability to quantify mRNA transcripts, the RPA is a useful tool for investigating expression of low abundant receptor genes and their regulation when only very small amounts of tissue are available. Furthermore, it is suitable for use in clinical and experimental studies in which accurate quantification of transcripts is necessary.
PMID: 7665215, UI: 95394504
Prog Growth Factor Res 1995; 6(2-4): 397-408
Circulating and ovarian IGF binding proteins: potential roles in normo-ovulatory cycles and in polycystic ovarian syndrome
Giudice LC, van Dessel HJ, Cataldo NA, Chandrasekher YA, Yap OW, Fauser BC
Department of Gynecology and Obstetrics, Stanford University Medical Center, CA 94305, USA.
IGFs function as co-gonadotropins in the ovary, facilitating steroidogenesis and follicle growth. IGFBP-1 to -5 are expressed in human ovary and mostly inhibit IGF action in in vitro ovarian cell culture systems. In the clinical disorder of polycystic ovarian syndrome (PCOS), which is characterized by hyperandrogenemia, polycystic ovaries and anovulation, follicles have a higher androgen: estradiol (A : E2) content and growth is arrested at the small antral stage. In the PCOS follicle, follicle stimulating hormone (FSH) and IGF levels are in the physiologic range, and even in the face of abundant androstenedione (AD) substrate, aromatase activity and E2 production are low. When PCOS granulosa are removed from their ovarian environment, they respond normally or hyperrespond to FSH. It has been postulated that an inhibitor of IGF's synergistic actions with FSH on aromatase activity may be one (or more) of the IGFBPs, which contributes to the arrested state of follicular development commonly observed in this disorder. High levels of IGFBP-2 and IGFBP-4 are present in follicular fluid (FF) from androgen-dominant follicles (FFa) from normally cycling women and in women with PCOS. This is in marked contrast to the near absence of these IGFBPs in estrogen-dominant FF (FFe), determined by Western ligand blotting. Regulation of granulosa-derived IGFBPs is effected by gonadotropins and insulin-like peptides. In addition, an IGFBP-4 metallo-serine protease is present in FFe, but not in FFa in ovaries from normally cycling women and those with PCOS, although the IGFBP-4 protease is present in PCOS follicles hyperstimulated for in vitro fertilization. Recent studies demonstrate that IGF-II in FFe is higher than in FFa' whereas IGF-I, IGFBP-3 and IGFBP-1 levels do not differ, underscoring the importance of local IGF-II production by the granulosa and the importance of IGFBP-4 and IGFBP-2 in regulation of IGF-II action within the follicle during its developmental pathway as an E2- or A-dominant follicle. In the androgen-treated female-to-male transsexual (TSX) model for PCOS, IGF-I, IGF-II, IGFBP-3 and IGFBP-1 levels do not differ.
Publication Types:
- Review
- Review, tutorial
PMID: 8817683, UI: 96414662
Hum Pathol 1994 Nov; 25(11): 1198-1204
Androgen receptor expression in human ovarian and uterine tissue of long-term androgen-treated transsexual women
Chadha S, Pache TD, Huikeshoven JM, Brinkmann AO, van der Kwast TH
Department of Pathology, Erasmus University Rotterdam, The Netherlands.
Androgen receptor (AR) modulation in human uteri and ovaries of long term androgen-treated transsexual female patients was investigated. Androgen receptor expression was evaluated immunohistochemically in the ovaries of 11 and the endometria and myometria of six androgen-treated transsexual female patients. This was compared with AR expression in the ovaries and uteri of premenopausal and postmenopausal women not receiving treatment and in 10 ovaries of female patients with polycystic ovarian disease (PCOD). In the normal ovaries germinal epithelium, granulosa cells of antral follicles, corpus luteum, and thecal and stromal cells exhibited moderate AR expression. The more intense and uniform staining of ovarian stroma of female transsexual patients and those of patients with PCOD compared with ovarian stroma of normal controls was most remarkable. This similarity in histology and distribution of ARs supports the hypothesis that PCOD is an androgen-mediated disorder. Immunostaining for ARs was only occasionally detectable in the uteri of premenopausal and postmenopausal women. In contrast, myometrial and endometrial stroma of the uteri of female transsexual patients displayed an intense and diffuse nuclear immunostaining, but glandular epithelia remained unstained. Western blot analysis of the ovaries and uterine myometrial tissue samples from transsexual female patients confirmed the presence of the 110-kd AR molecule. Because the androgen treatment of some transsexual female patients was discontinued 6 weeks before they underwent hysterosalpingo-oophorectomy, our data indicate a stable and persistent androgen-induced up-regulation of AR expression in ovaries.
PMID: 7959665, UI: 95048264
Arch Pathol Lab Med 1994 May; 118(5): 562-565
Prolactin-producing pituitary adenoma in a male-to-female transsexual patient with protracted oestrogen administration. A morphologic study
Kovacs K, Stefaneanu L, Ezzat S, Smyth HS
Department of Pathology, St Michael's Hospital, Toronto, Ontario, Canada.
Pituitary adenoma developed in a 33-year-old male-to-female transsexual patient who was given estrogen, starting at 16 years of age; the pituitary adenoma was surgically removed and studied by light microscopy, immunocytochemistry, and in situ hybridization. The adenoma cells were immunoreactive for prolactin, and exhibited a strong signal for prolactin and estrogen receptor messenger RNAs and a weak signal for dopamine receptor messenger RNA. The question of whether the development of an adenoma was incidental or was the direct effect of estrogen or whether it was mediated via other mechanisms, such as activation of growth factors or oncogenes or inhibition of tumor-suppressing genes or other genetic abnormalities, remained unresolved. The present case, which, to our knowledge, is the first to describe structural findings of a pituitary adenoma in a transsexual patient who was given estrogen, reinforces the view that protracted stimulation may play a role in the genesis of endocrine tumors.
PMID: 8192565, UI: 94250127
Clin Endocrinol (Oxf) 1994 May; 40(5): 595-601
Testosterone administration increases adrenal response to adrenocorticotrophin
Polderman KH, Gooren LJ, van der Veen EA
Department of Internal Medicine, Free University Hospital, Amsterdam, The Netherlands.
OBJECTIVE: Various studies have demonstrated coexistence of ovarian and adrenal hyperandrogenism in women. This study was designed to determine whether testosterone can increase the response of the adrenal gland to stimulation by adrenocorticotrophin (ACTH). DESIGN: Non-randomized intervention in a university teaching hospital. PATIENTS: Twenty androgen-treated female-to-male transsexual patients (10 ovariectomized and 10 non-ovariectomized) and 10 normal female controls. MEASUREMENTS: ACTH stimulation tests were performed in all subjects. Baseline values and the increase above baseline values after ACTH stimulation were assessed for cortisol and the adrenal androgens androstenedione, dehydroepiandrosterone and dehydroepiandrosterone-sulphate. RESULTS: Increases in levels of cortisol, androstenedione and dehydroepiandrosterone after administration of ACTH were greater in testosterone-treated transsexual patients than controls. CONCLUSION: We conclude that testosterone increases the response of the adrenal gland to stimulation by ACTH.
PMID: 8013140, UI: 94282949
J Pathol 1993 May; 170(1): 37-43
An immunohistochemical study of the long-term effects of androgen administration on female-to-male transsexual breast: a comparison with normal female breast and male breast showing gynaecomastia
Burgess HE, Shousha S
Department of Histopathology, Charing Cross and Westminister Medical School, London, U.K.
This study was aimed at assessing the effects of therapeutic long-term administration of androgens on normal human female breast. Sections from mastectomy specimens of 29 female-to-male transsexuals who had prolonged androgen administration prior to surgery were examined using routine light microscopy and immunohistochemical techniques. For comparison, sections from ten 'normal' female breast reduction mammoplasty specimens and ten cases of gynaecomastia were similarly examined. Haematoxylin and eosin-stained sections were assessed for the prevalence of elements of the normal breast and benign breast lesions. Immunoperoxidase techniques were performed to study the distribution of a variety of breast-associated antigens and receptors. The results were assessed semi-quantitatively. The prevalence of normal acini and ducts, fibrosis, cysts, and apocrine metaplasia in transsexual specimens was not statistically different from that seen in normal controls. However, transsexual specimens had a significantly higher prevalence of microcalcification than normals. The majority of transsexual specimens were positive for gross cystic disease fluid protein-15, lactoferrin, and progesterone and oestrogen receptors, and negative for B72.3 and pS2. These findings were not significantly different from those in normal controls. All ten gynaecomastia specimens were positive for oestrogen and progesterone receptors. The prevalence of oestrogen receptors was significantly higher than that seen in transsexuals and normal controls, but the prevalence of progesterone receptors was only significantly higher than that seen in transsexuals. It is concluded that long-term androgen administration does not appear to have any significant lasting effect on the normal human female breast, as demonstrated by a wide range of histological and immunohistological criteria.
PMID: 8326458, UI: 93316241
Ann Intern Med 1993 Mar 15; 118(6): 429-432
Influence of sex hormones on plasma endothelin levels
Polderman KH, Stehouwer CD, van Kamp GJ, Dekker GA, Verheugt FW, Gooren LJ
Free University Hospital, Amsterdam, The Netherlands.
OBJECTIVE: To determine whether a sex-associated difference in endothelin levels exists and to assess whether this difference is mediated by sex hormones. DESIGN: Initial survey in healthy persons, followed by a nonrandomized intervention. SETTING: A university teaching hospital. PATIENTS: Twenty-three healthy men, 29 healthy women, 20 pregnant women, and 12 male-to-female and 13 female-to-male transsexual patients. MEASUREMENTS: Plasma levels of endothelin were first assessed in healthy men, healthy women, and pregnant women; subsequently, endothelin levels were determined in male-to-female and female-to-male transsexual patients both before and during cross-gender hormone treatment. This treatment involves administration of testosterone esters to women and of ethynylestradiol and cyproterone acetate to men. RESULTS: Endothelin levels were higher in men than in women (5.9 +/- 1.2 compared with 4.17 +/- 0.67 pg/mL; P < 0.01). Endothelin levels were lower in pregnant women than in age- and sex-matched nonpregnant controls (2.19 +/- 0.73 compared with 4.17 +/- 0.67 pg/mL; P < 0.01). In 12 male-to-female transsexuals treated with estradiol and the progestational agent cyproterone acetate, endothelin levels decreased from 8.1 +/- 3.0 to 5.1 +/- 2.0 pg/mL (P < 0.01). In 13 female-to-male transsexuals treated with testosterone, endothelin levels increased from 6.2 +/- 1.1 to 7.8 +/- 1.2 pg/mL (P < 0.01). CONCLUSION: Sex hormones may modulate plasma endothelin levels, with male hormones raising levels and female hormones lowering them. This finding may be important in explaining sex-associated differences in susceptibility to atherosclerotic cardiovascular disease.
PMID: 8439117, UI: 93175793
Fertil Steril 1992 Sep; 58(3): 603-608
Effects of ethinyl estradiol on semen quality and various hormonal parameters in a eugonadal male
Lubbert H, Leo-Rossberg I, Hammerstein J
Hospital for Women, Department of Gynaecological Endocrinology and Reproductive Medicine, Free University of Berlin, Germany.
OBJECTIVE: To determine the influence of estrogens on male fertility. DESIGN: A 36-year-old eugonadal male was subjected to two different regimens of treatment with ethinyl estradiol (EE2). Sperm quality, immunoreactive luteinizing hormone (LH) and follicle-stimulating Hormone (FSH), testosterone (T), estrone (E1), estradiol (E2), dehydroepiandrosterone sulfate (DHEAS), prolactin (PRL) and sex hormone-binding globulin were determined at intervals of 2 weeks for 315 days. SETTING: A gender dysphoria clinic. PATIENT: A transsexual male nurse. MAIN OUTCOME MEASURES: It was hypothesized (and confirmed) that by comparing the effects of increasing and constant dose of EE2 on fertility parameters, differences in estrogen-sensitivity would show more clearly. Furthermore, this procedure served to find the minimal dose of EE2 for complete testicular suppression. RESULTS: Low doses of EE2 (20 micrograms/d) had no negative effect on sperm motility and density for a period of approximately 4 weeks, whereas high doses (60 micrograms/d) reduced motility already after a few days and led to a pronounced decrease in sperm density after 2 weeks. After discontinuation of therapy, motility normalized faster than sperm density. Under increasing doses of EE2 there was a constant decrease of FSH that occurred several weeks earlier than that of LH. Under constant dose of EE2 (60 micrograms/d) the decrease of LH was delayed (with respect to FSH) by only a few days. The decrease in T showed a stronger correlation with that of FSH than with that of LH. Volume and fructose content of the seminal fluid correlated with the decrease in T. Rebound effects were observed for FSH, LH, T, and fructose during the therapy-free interval. Ethinyl estradiol therapy had no influence on the serum concentrations of E1, E2, and PRL. Estrone was the dominant estrogen before and after therapy with EE2. Adrenal gland activity was markedly suppressed by EE2, as reflected by the decrease in DHEAS. CONCLUSION: The suppressive effect of EE2 on FSH and sperm motility was more pronounced and consistent than on LH and sperm density. The T decrease appears to be mainly caused by a direct effect of EE2 on the testes.
PMID: 1387850, UI: 92394314
Med Hypotheses 1992 Sep; 39(1): 85-87
Are all males equal? Anatomic and functional basis for sexual orientation in males
Elias AN, Valenta LJ
University of California, Irvine 92717.
In non-human primates anterior hypothalamic nuclei are closely involved in heterosexual activity in males. In humans, hypothalamic nuclei which correspond to these nuclei in non-human primates have been shown to have a neuronal density in homosexual men that is approximately half of that seen in heterosexual men. In addition, homosexual men exhibit a positive luteinizing hormone response to acutely administered estrogen that is intermediate between women (flat response) and heterosexual men (exaggerated response). Furthermore, on the basis of serum testosterone concentrations in similarly feminized transsexual males on estrogen/progestogen treatment 3 distinct groups can be identified. It is postulated that anatomic differences in the anterior hypothalamic nuclei that regulate sexual orientation in males may lead to alteration in the gonadotropin releasing hormone (GnRH) pulse/frequency leading to a more female-type pattern of gonadotropin secretion in homosexual males. Based on data in transsexual males this pattern may be more or less of the female-type in subsets of male homosexuals.
PMID: 1331721, UI: 93062278
Clin Endocrinol (Oxf) 1992 Jun; 36(6): 565-571
17 beta-Oestradiol, androstenedione and inhibin levels in fluid from individual follicles of normal and polycystic ovaries, and in ovaries from androgen treated female to male transsexuals
Pache TD, Hop WC, de Jong FH, Leerentveld RA, van Geldorp H, Van de Kamp TM, Gooren LJ, Fauser BC
Department of Obstetrics and Gynaecology, Dijkzigt University Hospital, Rotterdam, The Netherlands.
OBJECTIVE: The aim was to monitor carefully follicular growth arrest in polycystic ovaries by assay of hormones in individual follicles. DESIGN AND PATIENTS: Fluid from follicles less than or equal to 10 mm was obtained from ovaries of 16 regularly cycling women between days 1 and 12 of the follicular phase (controls, n = 120 follicles), polycystic ovaries of five women with polycystic ovary syndrome (n = 43), and polycystic ovaries from 14 long-term testosterone treated female to male transsexuals (n = 120). MEASUREMENTS: Fluid was assayed for oestradiol, androstenedione, and immunoactive inhibin. Luteinizing hormone, follicle-stimulating hormone, and testosterone levels were estimated in serum. RESULTS: Median serum LH was lower in transsexuals than in controls (P less than 0.05), and in polycystic ovary syndrome (P less than 0.01). Median serum testosterone was not significantly different between polycystic ovary syndrome and transsexuals, and was elevated in both groups as compared to controls (P less than 0.01). Oestradiol was present in all follicles obtained from polycystic ovaries of polycystic and transsexual patients, in which no follicle greater than 10 mm could be detected. In the three groups, between-patient differences in mean oestradiol, androstenedione, inhibin, and androstenedione/oestradiol ratio were significantly larger than expected in view of the variation between follicles within individuals. Taking into account this between-patient difference, no significant differences could be established between the three groups for all endocrine parameters. The percentage of presumed healthy follicles (androstenedione/oestradiol ratio less than or equal to 4) was 12% in controls, 17% in polycystics, and 14% in transsexuals, and was not significantly different between groups. CONCLUSIONS: The results may indicate that (1) abnormally high circulating androgen concentrations with or without elevated LH levels disturb the process of selection, and could therefore play a role in the pathogenesis of polycystic ovaries; (2) in polycystic ovaries from polycystic ovary syndrome and transsexual patients, aromatase activity is present in vivo in small antral follicles, and the proportion of presumed healthy follicles is not different from that encountered in normal ovaries; (3) oestradiol levels are not different between non-dominant follicles of normal and polycystic ovaries, suggesting that only enhancement of aromatase activity by FSH may be disrupted in polycystic ovaries, (4) because androstenedione levels are not different comparing follicles of normal and polycystic ovaries, hyperandrogenaemia in the syndrome seems to originate from the abnormally high number of cystic atretic follicles generally observed in polycystic ovaries; (5) marked variation in the endocrine follicular microenvironment within and between-women precludes pooling fluid from several follicles.
PMID: 1424181, UI: 93047364
J Natl Med Assoc 1992 Mar; 84(3): 241-250
Hormone pattern in pharmacologically feminized male transsexuals in the California State prison system
Valenta LJ, Elias AN, Domurat ES
Patton State Hospital, Department of Mental Health, State of California.
The hormonal profile of 40 transsexual inmates from a pool of 86 inmates in the California State prison system was studied before and after therapy with feminizing hormones. Clinical and social data were obtained on all 86 inmates; the incidence of human immunodeficiency virus (HIV) seropositivity was examined in 76 of the 86 individuals. Despite similar degrees of feminization in all 40 individuals in whom hormonal studies were performed, variable suppression of serum testosterone concentrations was present. Based on their testosterone concentrations while on feminizing hormone therapy, the transsexual inmates could be divided into three groups. In Group I (the "suppressed" group), the serum testosterone concentrations were markedly depressed (less than 10 ng/dL); in Group II (the "non-suppressed" group), the values of testosterone were normal (446 to 1072 ng/dL); and in Group III (the "intermediate" group), the testosterone values were between those of the suppressed group and the nonsuppressed group. We speculate that feminizing hormone therapy may induce the development of a state of target hormone resistance to testosterone that results in similar degrees of feminization independent of the circulating concentrations of testosterone. The incidence of HIV seropositivity (3/76) was considerably less than anticipated based on previous studies in populations at high risk for developing the acquired immunodeficiency syndrome.
PMID: 1578499, UI: 92251817
Arch Sex Behav 1990 Oct; 19(5): 507-516
Effect of estrogens on prolactin secretion in transsexual subjects
Goh HH, Ratnam SS
Department of Obstetrics and Gynecology, National University of Singapore, National University Hospital, Kent Ridge.
The effect of estrogens on the secretion of prolactin in 8 different groups of transsexual subjects was studied. Two different types of estrogens, estradiol or its conjugate and ethinyl estradiol, were used. Different doses and durations of exposure were employed. Plasma levels of prolactin and SHBG after estrogen exposure were compared with corresponding levels before treatment. Results showed that for estrogens to exert an enhancing effect on the secretion of prolactin, three factors needed to be considered: (i) the absolute concentration of estrogen, (ii) the duration of exposure, and (iii) whether levels of SHBG are sufficiently altered to change the concentration of free estrogen. It appears that there exist both time and dose thresholds for effective enhancement of prolactin secretion by estrogen. Estradiol and its conjugate are more likely to induce hyperprolactinemia than ethinyl estradiol. The reduced effect of the latter is probably related to its ability to induce large and rapid increases in SHBG binding which probably results in unaltered free estrogen concentration. In the light of this study, treatment of male transsexuals with high doses of estradiol in its conjugate forms must be viewed with caution.
PMID: 2260915, UI: 91083478
Gynecol Endocrinol 1990 Jun; 4(2): 127-141
Effects of estrogens, clomiphene and castration in a male transsexual with as compared to those without hypersecretion of gonadotropins
Goh HH, Ratnam SS
Department of Obstetrics and Gynaecology, National University Hospital, National University of Singapore.
A 24-year-old male-to-female transsexual with hypersecretion of gonadotropins was studied with regard to his recovery from sex steroid hormone treatment before his sex change operation and the effects of castration, clomiphene tests and estrogens on the hypersecretion of gonadotropins. Computer-assisted tomography and X-ray results indicated that the possibility that the hypersecretion of gonadotropins was not associated with a pituitary adenoma could not be ruled out. After a period of recovery from previous sex steroid hormone therapy, inappropriately high FSH and LH levels in the presence of normal male levels of testosterone and estradiol were found. The latter normal levels failed to suppress both the FSH and LH levels. From the clomiphene challenge tests carried out before and after the sex reassignment operation, the estradiol infusion studies and treatment with ethinyl estradiol after the sex reassignment operation, it appears that the hypersecretion of gonadotropins was responsive to the negative feedback effect of estrogens. However, the sensitivity, especially that of FSH, was very attenuated. Even after 23 weeks of ethinyl estradiol treatment FSH remained well above the upper limit for normal men. The results showed that before his sex change operation the hypersecretion of gonadotropins in the patient was probably under autonomous control. Testicular factors at normal male concentrations appear to have a minimal negative feedback effect on the hypersecretion of gonadotropins. However, hypersecretion of gonadotropins can be suppressed by prolonged exposure to estrogens.
PMID: 2118708, UI: 90372115
Arch Ital Urol Nefrol Androl 1990 Mar; 62(1): 121-123
Effects of long-term administration of androgens on the breast in transsexual women
[Article in Italian]
Godano A, Sapino A, Pietribiasi F, Bussolati G
Ospedale Mauriziano di Torino.
At present the effect on chronic hormonal treatment of normal mammary glands is not known. However, it is thought that high levels of androgens can cause both stromal sclerosis and epithelial atrophy and might provoke fibrocystic mastopathy. The authors describe their own histological studies performed on mammary glands of two women. They underwent mastectomy for transsexualism after prolonged treatment with testosterone.
PMID: 2141705, UI: 90296088
Arch Ital Urol Nefrol Androl 1990 Mar; 62(1): 107-111
Male transsexualism and hormonal therapy: radiologic pictures of the breast
[Article in Italian]
Godano A, Massara D, Grassi G, Genovese MG, Cavallotti GP, Bocchini R
Divisione di Ostetricia e Ginecologia, Ospedale Mauriziano, Torino.
Often the male transsexual takes hormonal drugs, sometimes under medical prescription but most frequently by his personal choice. They wish to obtain a development of mammary gland and the disappearance of hair from facial skin. Prolonged hormonal and incorrect treatments, mainly for too high dosage and absence of association with progesterone, assumption of oestrogens, can cause the growth of mammary tumours. The Authors present the results of a clinical and radiological study on 15 patients, 12 in treated with hormonal drugs for two years and 3 without any therapy.
PMID: 2141702, UI: 90296085
Br J Psychiatry 1990 Feb; 156: 285-286
Psychosis in a transsexual
Faulk M
Publication Types:
- Comment
- Letter
Comments:
- Comment on: Br J Psychiatry 1989 Aug;155:257-9
PMID: 2317639, UI: 90199389
Metabolism 1989 Sep; 38(9): 869-873
Mortality and morbidity in transsexual patients with cross-gender hormone treatment
Asscheman H, Gooren LJ, Eklund PL
Department of Internal Medicine, Free University Hospital, Amsterdam, The Netherlands.
Sex steroid treatment is associated with side effects. The number of deaths and morbidity cases in 425 transsexual patients treated with cross-gender hormones were evaluated retrospectively and compared with the expected number in a similar reference group of the population. The number of deaths in male-to-female transsexuals was five times the number expected, due to increased numbers of suicide and death of unknown cause. Combined treatment with estrogen and cyproterone acetate in 303 male-to-female transsexuals was associated with a 45-fold increase of thromboembolic events, hyperprolactinemia (400-fold), depressive mood changes (15-fold), and transient elevation of liver enzymes. Androgen treatment in 122 female-to-male transsexuals was associated with weight increase greater than 10% (17.2%) and acne (12.3%). In both groups persistent liver enzyme abnormalities could be attributed to other causes than sex steroids (hepatitis B and alcohol abuse). Much of the morbidity was minor and reversible with appropriate treatment or temporary discontinuation of hormone treatment. Thus, the dilemma of prescribing cross gender hormones in view of the needs of these patients is not resolved. Explanation of possible side effects and careful clinical judgment remain the cornerstone of the clinical decision to prescribe cross-gender hormones. Furthermore, follow up of this relatively young population to disclose long-term side effects and to elucidate the association of sex steroids with coronary heart disease, as well as efforts to reduce the risk of thromboembolic events, are required.
PMID: 2528051, UI: 89364275
J Endocrinol Invest 1989 Jul; 12(7): 449-454
Exogenous estrogen effect on lipid/lipoprotein cholesterol in transsexual males
Damewood MD, Bellantoni JJ, Bachorik PS, Kimball AW Jr, Rock JA
Department of Gynecology and Obstetrics, Johns Hopkins University School of Medicine, Baltimore, Maryland 21205.
Lipid/lipoprotein cholesterol values and sex-hormone-binding globulin levels were determined in 40 transsexual males aged 20-38, 20 castrated and 20 non-castrated, taking conjugated estrogens to induce female characteristics. Variables controlled included dose of estrogen, age, weight, smoking, alcohol intake, exercise and diet history. Transsexual males on estrogens had significantly higher mean (+/- SE) HDL cholesterol levels (69.0 +/- 7.1 mg/dl) respectively, for castrated males and (53.8 +/- 6.2 mg/dl) for non-castrated males, respectively compared to normal control males not on hormonal therapy (41.5 +/- 5.4) (p less than 0.001), regardless of dose of estrogen received. The total cholesterol/HDL ratio was 3.31-4.05 in transsexual males on estrogens compared to 5.03 for normal males (p less than 0.001). Transsexual males had mean SHBG levels in the female range (63.4 to 71.8 nmol/ml), significantly higher than controls (26.7 nmol/ml) (p less than 0.001). SHBG levels were correlated with estrogen use, dose and HDL cholesterol levels. We conclude that exogenous estrogens administered to transsexual males results in a female pattern of lipid/lipoprotein cholesterol and SHBG concentration. The decreased total cholesterol/HDL ratio may imply a lower atherogenic potential and a lessened cardiovascular risk in males who take estrogens.
PMID: 2794371, UI: 90009891
J Clin Endocrinol Metab 1989 Jul; 69(1): 151-157
The effects of long term testosterone administration on pulsatile luteinizing hormone secretion and on ovarian histology in eugonadal female to male transsexual subjects
Spinder T, Spijkstra JJ, van den Tweel JG, Burger CW, van Kessel H, Hompes PG, Gooren LJ
Department of Internal Medicine, Free University Hospital, Amsterdam, The Netherlands.
Polycystic ovarian disease (PCOD) is associated with elevated serum LH and (sub)normal FSH levels, while serum androgen levels are often elevated. To clarify the role of androgens in this abnormal pattern of gonadotropin secretion, LH secretion was studied in 1) 9 eugonadal female to male transsexual subjects before and during long term (6 months) testosterone (T) administration (250 mg/2 weeks, im), and 2) in a woman with an androgen-secreting ovarian tumor both before and after surgical removal of the tumor. Finally, we studied the effects of high serum androgen levels on ovarian histology in 3) 26 transsexual subjects after long term (9-36 months) T administration (250 mg/2 weeks, im) to assess whether T-induced ovarian abnormalities are similar to those that occur in women with PCOD. Long term T treatment in the nine female to male transsexual subjects resulted in increases in the mean serum T level from 1.7 +/- 0.8 (+/- SD) to 40.8 +/- 31.9 nmol/L (P less than 0.01), the mean serum dihydrotestosterone level from 0.6 +/- 0.2 to 3.3 +/- 1.5 nmol/L (P less than 0.02), and the mean serum free T level from 9.5 +/- 5.2 to 149 +/- 46 pmol/L (P less than 0.02). Mean serum estrone and estradiol levels were similar before and during T treatment. The mean serum LH level decreased from 6.3 +/- 2.0 to 2.9 +/- 1.1 U/L (P less than 0.01), and the mean FSH levels decreased from 6.6 +/- 2.0 to 3.7 +/- 2.2 U/L (P less than 0.02). Pulsatile LH secretion before and during T treatment was studied in five subjects. Neither the mean nadir LH interval nor the LH pulse amplitude changed significantly in these five subjects. The serum T level in the woman with the androgen-secreting ovarian tumor was 9.6 nmol/L, and it declined to normal after removal of the tumor. Her mean serum LH and FSH levels, the mean nadir LH interval, and LH pulse amplitude were in the normal range before and after removal of the tumor. Studies of ovarian histopathology in 26 transsexual subjects after long term androgen treatment revealed multiple cystic follicles in 18 subjects (69.2%), diffuse ovarian stromal hyperplasia in 21 subjects (80.8%), collagenization of the tunica albuginea in 25 subjects (96.2%), and luteinization of stromal cells in 7 subjects (26.9%). Findings consistent with criteria for the pathological diagnosis of polycystic ovaries, that is 3 of the 4 findings listed above, were present in 18 of the 26 subjects (69.2%).
PMID: 2471710, UI: 89278278
Arch Sex Behav 1989 Feb; 18(1): 49-57
Spironolactone with physiological female steroids for presurgical therapy of male-to-female transsexualism
Prior JC, Vigna YM, Watson D
Department of Medicine (Endocrinology), University of British Columbia, Vancouver General Hospital, Canada.
The clinical and hormonal response to 12-month therapy with the antiandrogen, spironolactone, in conjunction with near-physiologic doses of female gonadal steroids in 50 transsexual males, is presented. An unselected referred series of 61 men with the psychiatric diagnosis of transsexualism was treated; 10 subjects who had received previous gonadal surgery and 1 man with Klinefelter's syndrome were excluded. Twenty-seven conventionally treated (CT; high-dose estrogen), age 34.4 +/- 10.5 years, mean +/- SD, and 23 untreated patients (SPS), age 30.7 +/- 6.2 years, were studied. Following the initial visit, all 50 were begun on spironolactone and low-dose female hormone therapy. Despite high-dose estrogen treatment for more than 2 years, the mean testosterone (T) level for the CT group was not in the female range (169 +/- 193 ng/dl; normal 20-80). Spironolactone, in doses of 200-600 mg/day, lowered T to the female range in both groups after 12 months (CT 87 +/- 111 and SPS 49 +/- 41 ng/dl). This was achieved in the CT group despite decreases in estrogen dose and discontinuation of parenteral therapy. SPS subjects experienced significant decreases in plasma T (642 +/- 236 to 49 +/- 41 ng/dl, p less than 0.001). Systolic blood pressure dropped (128 +/- 14 to 121 +/- 14 mm Hg, p less than 0.05). The clinical response, including decreased male pattern hair, breast development, feminization, and lack of erections was excellent in most subjects.
PMID: 2540730, UI: 89227588
Sb Lek 1989 Jan; 91(1): 16-20
Comparison of the excretion of 3 androgens registered in Czechoslovakia
[Article in Czech]
Hampl R, Hana V, Heresova J, Brzek A, Marek J, Starka L
Hypogonadal men of different aetiology were administered testesterone undecanoate (Andriol, Organon) and methyltestosterone (Agovirin coated tablets, Spofa) for three months alternatively. By means of specific radioimmunoassays excretion of testosterone and methyltestosterone respectively in the urine was observed in 20 and 5 patients respectively. The course of excretion of both the preparations is possible to illustrate by decreasing curves with apparent half-lives 1.74 and 3.90 day respectively. Analogous observation of testosterone excretion after three-month administration of testosterone isobutyrate (Agovirin depot) to five female-to-male transsexual patients was observed. Excretion course shows the maximum on 27th day after the treatment was discontinued and it is not possible to illustrate it by a monotonous curve.
PMID: 2717876, UI: 89241913
Psychoneuroendocrinology 1989; 14(1-2): 97-102
Pulsatile luteinizing hormone release and ovarian steroid levels in female-to-male transsexuals compared to heterosexual women
Spinder T, Spijkstra JJ, Gooren LJ, Burger CW
Department of Internal Medicine, Free University Hospital, Amsterdam, The Netherlands.
It has been suggested that there is a difference in the interaction between sex steroids and gonadotropin secretion between transsexual and heterosexual women. In this study we tested whether there is a difference in the pulsatile release characteristics of luteinizing hormone (LH) and in ovarian steroid concentrations between 16 female-to-male (f-to-m) transsexuals and eight eugonadal heterosexual women during the early follicular phase of the menstrual cycle. Blood samples for LH determination were collected every 10 min for 7 hr in the transsexual group, and every 10 min for 6 hr in the heterosexual group. There were no significant differences between the transsexual and heterosexual group in mean serum LH concentration (6.9 +/- 2.4 U/l vs. 7.5 +/- 2.6 U/l), mean LH nadir interval (77 +/- 37 min vs. 70 +/- 16 min), and mean LH pulse amplitude (2.9 +/- 1.1 U/l vs. 3.0 +/- 1.1 U/l). Serum estrone, 17-beta-estradiol, testosterone, progesterone and FSH concentrations also did not differ between the two groups. There also was not a higher prevalence of polycystic ovarian disease in our f-to-m transsexual group than in the general population.
PMID: 2499904, UI: 89283123
Clin Plast Surg 1988 Jul; 15(3): 471-487
Transsexual surgery in the genetic female
Gilbert DA, Winslow BH, Gilbert DM, Jordan GH, Horton CE
Department of Plastic Surgery, Eastern Virginia Medical School, Norfolk.
The increasing acceptance of the transsexual patient by society as well as a willingness of insurance companies to compensate for associated surgical procedures will result in an increase in patients presenting for sex reassignment. Data from gender identity centers suggest that the multidisciplinary approach leads to the best therapeutic results. Significant advances in phalloplasty techniques are yielding genitalia with more normal function and appearance.
Publication Types:
- Review
- Review, tutorial
PMID: 3292116, UI: 88270842
Books
Amazon.com is pleased to have Confluence Publications in the family of Amazon.com associates. We've agreed to ship books and provide customer service for orders we receive through special links from Confluence Publications.
Amazon.com associates list selected books in an editorial context that helps you choose the right books. We encourage you to visit Confluence Publications often to see what new books they've selected for you.
Thank you for shopping with an Amazon.com associate.
Jeff Bezos President Amazon.com Books
P.S. We guarantee you the same high level of customer service you would receive at Amazon.com. If you have a question about an order you've placed, please don't hesitate to contact us.
Basic Endocrinology
Dr. Susan Love's Hormone Book: Making Informed Choices about Menopause http://www.amazon.com/exec/obidos/ISBN=081296392X/confluencepublicA/ by Susan Love M.D. Dr. Susan Love, one of the most important amd reputable doctors addressing women's issues today, helps readers sort through the confusing media reports, complex and contradictory scientific evidence, and medical bias regarding hormone therapy, discussing what is known and not known about it as well as alternatives.
Endocrinology http://www.amazon.com/exec/obidos/ISBN=0133179265/confluencepublicA/ by Mac Hadley Hadley provides an comprehensive coverage of endocrinology, centralizing on the critical roles of chemical messengers and hormones--whether they are of endocrine or neural origin--in the control of physiological processes. This up-t o-date Fourth Edition depicts, through demonstration, the entire human endocrine system in examples designed specifically for premedical and related professional courses. Hadley examines several recently discovered hormones--including their origin , biosynthesis, chemistry, secretion, circulation and metabolism, physiological roles, control of secretion, mechanisms of action and pathophysiology.
Estrogen http://www.amazon.com/exec/obidos/ISBN=0060926023/confluencepublicA/ by Lila Nachtigall, Joan Rattner Heilman Highlighting new, groundbreaking discoveries in hormone replacement therapy's benefits, the nation's foremost authority offers a complete examination of this popular treatment used to counter the negative effects of menopause.
The Estrogen Alternative: Natural Hormone Therapy With Botanical Progesterone http://www.amazon.com/exec/obidos/ISBN=0892816457/confluencepublicA/ by John Lee, Judi Gerstun, Raquel Martin Sylvia Crawley, M.D. reviews The Estrogen Alternative saying "As women become more and more involved in decisions about hormone therapy, this book "serves a vital need. It is very timely and addresses this increasinly complex problem. The dilemma is due, in great part, to the many inconclusive and contradictory studies pubished by traditional medical institutions. It examaines the benefits of natural progesterone supplementation for women of all ages, with the author's shared experiences making THE ESTROGEN ALTERNATIVE even more appealing. She offers educational support for women who wish to participate actively in decisions about their own care. She also presents a challenge to the physicians who, it is hoped, will become more receptive to patients wishing to consider a more natural approach." Joseph A. Randall, M.D. states that this book "has been instrumental in changing the health care I provide to women. Where once I focused on synthetic HRT with estrogen, thinking the benefits outweighed the risks, I now strongly support the use of natural HRT. For example, the book documents the fact that estrogen replacement merely delays but does not prevent osteoporosis. Natural progesterone, with its virtual absnce of side effects, makes sense; and using it in my practice (OB/GYN and fertility) has greatly improved my patient's quality of life."
Estrogen and Breast Cancer: A Warning to Women http://www.amazon.com/exec/obidos/ISBN=089793198X/confluencepublicA/ by Carol Ann Rinzler A statistics-laden, fact-filled chronicle linking the increasing use of estrogen with the growing incidence of breast cancer, plus some solid ideas about remedying the situation. Medical writer Rinzler (Cosmetics, 1977, etc.) adeptly wends her way through a host of scientific studies, translating them into lay language and sorting out their implications. Her take- home message: The estrogen in oral contraceptives, used by millions of young women, and in hormone-replacement therapy, used by millions of older women, promotes the growth of existing tumors and may initiate cancers in susceptible women. Rinzler's story essentially begins with legal approval of the Pill in 1960, but there's also a brief look at how female medical problems were treated in earlier days, including some fascinating medical lore- -e.g., that in 1934, the Merck Manual, a standard reference book of current medical opinion, listed cannabis as a treatment for the symptoms of menopause. Rinzler generally lets the facts speak for themselves, offering no shrill diatribe against pharmaceutical companies or physicians, no easy indictment of the medical establishment. The picture she creates is more complex, since powerful medications such as estrogen are never risk-free, and the benefits always must be weighed against the dangers. Overall, Rinzler's criticisms are validated and her recommendations restrained: Women must be told the truth about estrogen, and those at risk for cancer must be identified; the rules for prescribing estrogen must be tightened; safer alternatives must be found. If the author meets her aim, women won't abandon the Pill or hormone-replacement therapy, but will ask critical questions of their physicians and make informed decisions about the risks they're willing to take. Straight talk--informative and accessible--about a health issue of concern to millions.
The Hormone of Desire: The Truth About Sexuality, Menopause, and Testosterone http://www.amazon.com/exec/obidos/ISBN=0517703424/confluencepublicA/ by Susan Rako "Conditions of testosterone deficiency do exist, need attention, and can be treated," Rako maintains as she draws on her practical experience as a psychiatrist as well as her thorough knowledge of relevant medical literature to discuss the usefulness of small doses of testosterone in helping women through menopause and stimulating positive sexual and psychological feelings. Women produce some testosterone just as men produce some estrogen. Yet many physicians, especially male gynecologists and endocrinologists, believe that giving testosterone to women is unnatural and potentially dangerous. Rako points out, however, that women do not display a simple dose-response curve for such treatment. This means that physicians not only have to overcome feelings of unnaturalness if they wish to aid some of their female patients but also must take the time and effort to match dosage to each individual's need.
Natural Woman, Natural Menopause : Complete Program for Healthy Menopause http://www.amazon.com/exec/obidos/ISBN=0060173416/confluencepublicA/ by Marcus Laux, Christine Conrad Accessible as well as authoritative, Natural Woman, Natural Menopause features the stories of other women who, like Christine Conrad, found that they didn't have to accept less than a completely safe alternative. More important, in Natural Woman, Natural Menopause Marcus Laux and Christine Conrad offer readers their complete plan for long-lasting health and renewed vitality. Following their "Natural Woman Plan," which features the right combination of plant-derived hormones, nutritional supplements, a plant-rich diet, and an exercise program to eliminate and even reverse the effects of bone loss, women will find that not only will they be more energetic and radiant than they thought possible but they will be adding years of good health to their lives.
Advanced Endocrinology
Technical references are generally expensive and special-order. If you are interested, you can search for all books on endocrinology.
Of General Interest to Transsexuals
Body Alchemy : Photographs http://www.amazon.com/exec/obidos/ISBN=1573440620/confluencepublicA/ by Loren Cameron Body Alchemy: Transsexual Portraits is photographer Loren Cameron's intensely personal photo documentary of female-to-male transsexuals (FTMs). A transsexual himself, Loren Cameron brings a sensitive, sophisticated insider's eye to his subject matter. Using documentary style, a series of before-and-after photographs of FTMs in Cameron's transsexual community, his own striking self-portraits, and intimate autobiographical text, he invites the viewer to experience this transformational rite of passage. Body Alchemy includes intimate, narrative photographs of Loren and his partner, Kayt, a lesbian-identified woman whose relationship to Cameron affords her much to say about the fluidity of gender and queer identity. Finally, Body Alchemy includes photographs of genital reconstructions, accompanied by text from three anonymous FTMs who discuss how they feel about their surgeries. Andy Warhol, Robert Mapplethorpe, Dianne Arbus among many others have all trained their lenses on the transgendered figure. Never have the transgendered seriously photographed their own. Not until Loren Cameron, that is.
Conundrum http://www.amazon.com/exec/obidos/ISBN=0805003614/confluencepublicA/ by Jan Morris
Ftm : Female-To-Male Transsexuals in Society http://www.amazon.com/exec/obidos/ISBN=0253336317/confluencepublicA/ by Holly Devor Holly Devor spent many years compiling indepth interviews and researching the lives of transsexual and transgendered people, many of whom became her friends. She traces the everyday and significant events that coalesce in transsexual identity, culminating in gender and sex transformation. After an introduction which grounds the discussion in historical and theoretical contexts, the author takes a life course approach to understanding female-to-male transsexualism. Using her subjects' own words as illustrations, Devor looks at how childhood, adolescent, and adult experiences with family members, peers, and lovers work to shape and clarify female-to-male transsexuals' images of themselves as people who should be men.
Gender Outlaw: On Men, Women, and the Rest of Us http://www.amazon.com/exec/obidos/ISBN=0679757015/confluencepublicA/ by Kate Bornstein Kate Bornstein has been through some changes--a former heterosexual male, one-time Scientologist and IBM salesperson, now a lesbian woman writer and actress. In this work, she covers everything readers want to know about gender (but are too confused to ask) and takes a witty, incisive look at the radical new politics of sexuality and gender. Also includes Bornstein's play, Hidden: A Gender
In Search of Eve: Transsexual Rites of Passage http://www.amazon.com/exec/obidos/ISBN=0897891155/confluencepublicA/ by Anne Bolin In Search of Eve is an absorbing account of the sociocultural aspects of gender transition. . . . [Bolin] has produced a carefully crafted, clearly written monograph which scholars of both sexuality and gender can profitably read. I would recommend it also for upper-level students in such courses. The book contains many fascinating insights and new findings.
Sex Changes : The Politics of Transgenderism http://www.amazon.com/exec/obidos/ISBN=1573440728/confluencepublicA/ by Pat Califia Pat Califia needs no introduction for lesbian and gay readers. Her writings on sexuality, pornography, censorship, S/M, and other controversial topics have earned her the reputation of a fearless defender of the rights of sexual minorities -- and a fearless intellectual adversary. Sex Changes: The Politics of Transgenderism is Califia's honest, meticulously researched analysis of the contemporary history of transsexuality. Based on in-depth interviews with gender transgressors who "opened their lives, minds, hearts, and bedrooms to the gaze of strangers," Sex Changes demonstrates Pat Califia's hallmark candor and insight. Writing about both male-to-female and female-to-male transsexuals, Califia examines the lives of early transgender pioneers like Christine Jorgenson, Jan Morris, Renee Richards and Mark Rees; partners of transgendered people like Minnie Bruce Pratt; and contemporary transgender activists like Leslie Feinberg and Kate Bornstein. Includes bibliography, resources, index. Pat Califia is well-known as a sharp critic of repressive American attitudes toward gender, sexuality and pornography. She is the author of many books, including Public Sex: The Culture of Radical Sex. Califia lives in San Francisco, where she works as a therapist, primarily serving the gay/lesbian and transgendered communities.
Read My Lips : Sexual Subversion and the End of Gender http://www.amazon.com/exec/obidos/ISBN=1563410907/confluencepublicA/ by Riki Anne Wilchins Over the course of the past decade transgender politics have become the cutting edge of sexual liberation. While the sexual and political freedom of homosexuals has yet to be fully secured, questions of who is sleeping with whom pale in the face of the battle by transgender activists to dismantle the idea of what it means to be a man or a woman. Riki Anne Wilchins's Read My Lips is a passionate, witty, and extraordinarily intelligent look at how society not only creates men and women--ignoring the fluidity of maleness and femaleness in most people--but also explains how those categories generate crisis for most individuals. It is impossible to read Wilchins's ideas and not be provoked in fundamental and mysterious ways.
Transgender Care : Recommended Guidelines, Practical Inforrmation, and Personal Accounts http://www.amazon.com/exec/obidos/ISBN=1566395712/confluencepublicA/ by Gianna Israel, Donald Tarver By empowering clients to be well-informed medical consumers and by delivering care providers from the straitjacket of inadequate diagnostic standards and stereotypes, this book sets out to transform the nature of transgender care. In an accessible style, Gianna Israel and Donald Tarver discuss the key mental health issues, with much attention to the vexed relationship between professionals and clients. They propose a new professional role, that of "Gender Specialist." The book contains a wealth of practical information and accounts of people's experiences about coming out to one's employer or to one's friends or spouse. Several essays spell out the legal rights of transgender people with regard to insurance, work, marriage, and the use of rest rooms. The second part of the book consists of thirteen essays on a range of controversial topics.
Transsexuals: Candid Answers to Private Questions http://www.amazon.com/exec/obidos/ISBN=0895947900/confluencepublicA/ by Ramsey Gerald, Phd This is a great resource book for anyone trying to decide if they are really male-to-female or not. For those getting ready to step out of the closet this book will likely answer many of the questions your going to be asked by family and friends. It is also loaded with facts and figures, a detailed description of what to expect when going to a Gender Identity committee for the first time, and a complete listing of the Standards of Care by the Harry Benjamin International Gender Dysphoria Association, Inc.
True Selves : Understanding Transsexualism for Families, Friends, Coworkers, and Helping Professionals http://www.amazon.com/exec/obidos/ISBN=0787902713/confluencepublicA/ by Mildred Brown, Chloe Rounsley What is it like to grow up in the wrong body? Are transsexuals considered homosexuals? Filled with real-life stories, actual letters, and touching poems, True Selves paints a heartfelt portrait of the risk-taking, confusion, and--ultimately--the courage that transsexuals face as they struggle to reveal their true being to themselves and to others.
Other Web Sites
Bajamar Women's Healthcare Pharmacy http://www.thorcomp.com/bajamar/
Gonadotrope Cytochemistry http://cellbio.utmb.edu/childs/gon1.htm by Gwen Childs, PhD
Hormones and Heart Disease http://www.primenet.com/~beckster/hormheart.html by Rebecca Anne Allison, MD
Medline Search http://www.ncbi.nlm.nih.gov/Entrez/medline.html by the National Institute of Health
Menopausal Hormone Replacement Therapy and Cancer Risk http://imsdd.meb.uni-bonn.de/cancernet/600310.html by the National Cancer Institute
RXMed http://www.rxmed.com/
[Scalp Hair] Regrowth Reference http://www.regrowth.com/reference/ by John Ertel
Standards of Care http://www.symposion.com/ijt/ijtc0405.htm by the Harry Benjamin International Gender Dysphoria Association
Transsexual Women's Resources: Hormone Therapy http://www.mindspring.com/~alawrence/hormone by Anne Lawrence, MD
U.S. FDA Personal Use Import Policy http://www.ceri.com/import.htm by the Cognitive Enhancement Research Institute
United States Pharmacopeia http://www.usp.org
Women's International Pharmacy http://www.wipws.com/
Comments
comments powered by Disqus